LAWSUITS NEWS & LEGAL INFORMATION
Legal News Articles & Interviews
The Importance of Due Diligence During a Divorce
 April 26, 2024. By LAS Staff Writer.
April 26, 2024. By LAS Staff Writer.Surgeons Warned to Stop Using Synovo Total Hip Implants
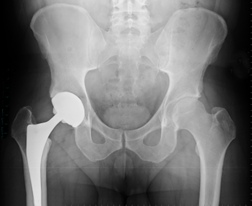 April 24, 2024. By Anne Wallace.
April 24, 2024. By Anne Wallace.Maryland Orders Employer to Fork Over Missed Contributions to 401k Plan
 April 22, 2024. By Anne Wallace.
April 22, 2024. By Anne Wallace.Bard PowerPort Bellwether Trial Process on Time
 April 19, 2024. By Jane Mundy.
April 19, 2024. By Jane Mundy.Ozempic Babies, Another Side Effect
 April 17, 2024. By Jane Mundy.
April 17, 2024. By Jane Mundy.Plaintiff calls for Appeal after Monsanto's Roundup Win in Philadelphia
 April 15, 2024. By Jane Mundy.
April 15, 2024. By Jane Mundy.Tesla Racism Lawsuit Settles for $3.2 million
 April 12, 2024. By Jane Mundy.
April 12, 2024. By Jane Mundy.No More Free Security Checks in California
 April 10, 2024. By Jane Mundy.
April 10, 2024. By Jane Mundy.Phillips 66 in $46.5 million Wage Dispute
 April 8, 2024. By Jane Mundy.
April 8, 2024. By Jane Mundy.Johnson & Johnson Strikes Back
 April 5, 2024. By Anne Wallace.
April 5, 2024. By Anne Wallace.- Hair Relaxer Lawsuit Update By Jane Mundy (Apr-3-24)
- 5 Effective Drug Possession Defense Strategies By LAS Staff Writer (Apr-2-24)
- Disneyland Not Happy Place for Maintenance Workers By Jane Mundy (Apr-1-24)
- Gig Economy Company Settles Wage Theft Lawsuit for $2.1 Million By Jane Mundy (Mar-27-24)
- Exactech Defective Hip Implant MDL Takes Shape By Anne Wallace (Mar-25-24)
- Meat Processors Pay $127.2 Million to Settle Wage-Fixing Case By Jane Mundy (Mar-20-24)
- DOL Charges TPA Embezzled ERISA Plan Assets By Anne Wallace (Mar-18-24)
- Ozempic MDL and Semaglutide Update By Jane Mundy (Mar-15-24)
- Bayer in the Weeds over Monsanto’s Roundup By Jane Mundy (Mar-13-24)
- 9 Best Ways to Remain Informed About Your Personal Injury Case By LAS Staff Writer (Mar-12-24)
- Glass House Slapped with California Labor Lawsuit By Jane Mundy (Mar-11-24)
- Lawsuits Allege Ozempic Caused Gastroparesis; Do You Have A Claim? By Jane Mundy (Mar-8-24)
- Former X Corp Workers ask Court not to Dismiss Gender Discrimination Lawsuit By Jane Mundy (Mar-7-24)
- California Employment Lawyer Discusses Wage & Hour Complaints By Jane Mundy (Mar-5-24)
- 44 Million Americans Drink PFAS-Contaminated Water, Says New Study By Jane Mundy (Mar-4-24)
- California Drywall Company Slapped with Wage & Hour Lawsuit – Again. By Jane Mundy (Mar-3-24)
- Court Won’t Dismiss Doctor’s Bayer Roundup Lawsuit By Jane Mundy (Feb-29-24)
- Last Dance, Last Chance for the Texas Two-Step in J&J Talc Litigation By Anne Wallace (Feb-28-24)
- Stryker Overtime Lawsuit Goes Ahead By Jane Mundy (Feb-26-24)
- Chicago Hospital Settles Class Action for $55M By Jane Mundy (Feb-22-24)
- PAGA To Face Critical Test on November 2024 Ballot By Anne Wallace (Feb-20-24)
- Things to Keep Handy When Consulting a Car Accident Injury Attorney By LAS Staff Writer (Feb-19-24)
- Suboxone maker Indivior in Trouble with the Law Again By Jane Mundy (Feb-16-24)
- Chemical Exposure Lawsuits By LAS Staff Writer (Feb-15-24)
- 3M Sending Payments for Faulty Combat Earplugs By Jane Mundy (Feb-12-24)
- New York MDL Consolidates Growing Number of Exactech Defective Hip Implant Lawsuits By Anne Wallace (Feb-9-24)
- Walmart Workers ask California Judge to OK $2.25M settlement By Jane Mundy (Feb-8-24)
- Another Option for Workers Who Cannot Save through an ERISA Retirement Plan By Anne Wallace (Feb-8-24)
- Delta Slammed with Lawsuit over Personal Cellphone Use By Jane Mundy (Feb-5-24)
- Two Popular California Bakeries Slammed with Wage Theft Lawsuits By Jane Mundy (Feb-1-24)
- Another Monsanto Roundup Loss for Bayer By Jane Mundy (Jan-31-24)
- The Cheesecake Factory Settles California Janitors Wage Theft Lawsuit By Jane Mundy (Jan-29-24)
- Weight Loss Drugmakers Face Stomach Paralysis Lawsuits By Jane Mundy (Jan-27-24)
- Nurses Filed Unpaid Wages Lawsuit Against U.S. Nursing Corp By Jane Mundy (Jan-25-24)
- Johnson & Johnson to Pay $700 Million to Settle State AG Lawsuits By Anne Wallace (Jan-23-24)
- More Long-Term Part Timers May Now Save Through 401k ERISA Plans By Anne Wallace (Jan-22-24)
- $120M awarded in Westchester Medical Center Medical Malpractice Lawsuit By Jane Mundy (Jan-21-24)
- UNITE HERE Dispute with Sky Chefs Back on the Plate By Jane Mundy (Jan-18-24)
- Hair Relaxers – Read the Label By Jane Mundy (Jan-16-24)
- Race Bias Lawsuit: Tesla v. EEOC By Jane Mundy (Jan-13-24)
