Posts Tagged ‘ Risperdal ’
Boy with Breasts? J&J Settles Risperdal Gynecomastia Lawsuit on 1st Day of Trial
September 10th, 2012. By AbiK
 Usually gynecomastia is a subject for plastic surgeons—not little boys on an antipsychotic drug like Risperdal.
Usually gynecomastia is a subject for plastic surgeons—not little boys on an antipsychotic drug like Risperdal.
However, a Risperdal lawsuit brought against Johnson & Johnson by Aron Banks was about just that—Banks alleged that by taking Risperdal, starting when he was just nine years old, he developed breast tissue (“gynecomastia” is the medical term) which led to sustained psychological trauma.
Banks, who is now 21, claimed he took Risperdal over a four-year period (2000-2004). The drug at the time had not been approved for children.
According to a Bloomberg report (9/10/12), Johnson & Johnson has settled Banks’ lawsuit on the opening day of trial in Philadelphia; another Risperdal lawsuit alleging gynecomastia is due to head to court in Philly on September 20.
Another drug that’s been linked to reports of gynecomastia is Propecia; currently, there are lawsuits underway alleging Propecia-induced sexual dysfunction.
Risperdal has also been linked to an increase in stroke risk for older patients who suffer dementia and take antipsychotics.
The terms of Banks’ Risperdal gynecomastia settlement have not been disclosed.
Forced Drugging of Kids Needs to Stop
June 23rd, 2011. By AbiK
Sometimes a child’s voice can be the voice of sanity and the impetus for change in the fight against a seemingly overwhelming foe. In this case, the foe is the mandated psychiatric drugging of our children with any of a number of drugs—Ritalin, Adderall, Risperdal to name a few. And the child, is a high school senior whose comment I came across on the Ablechild.org Facebook page.
The young man’s comment said that he has a nephew who is being forced to take medication—and that his nephew’s mother runs the risk of getting “screwed” should she not give the child the meds. He goes on to mention a friend who’s been on Ritalin his whole life and is “following a pathway to drug addiction and it is happening before my eyes.”
Given the recent study in the Journal of the American Academy of Child & Adolescent Psychiatry showing that childhood attention deficit hyperactivity disorder (ADHD) increases the risk that a child will smoke cigarettes or abuse drugs or alcohol in early adulthood, and that intervention with stimulant drugs was not found to conclusively alter or reduce that risk, either this kid has done his homework and/or he’s seeing a medical experiment play out in front of his very eyes.
Regardless, the comment he made that really caught my eye and stuck with me the most was this: “I wanna do something to stop this but I don’t know where to start I’m just a senior in high school.”
Just a senior in high school.
As someone who’s now well beyond high school and a parent to three, it’s a heart-wrenching plea to hear from someone so young, and clearly not so naive. Heart-wrenching because it’s compounded by the advice of the Ablechild.org FB page moderator to take advantage of the Hatch Amendment and the Medwatch form for reporting any adverse drug reactions to the FDA. Yes, a parent should be aware of both and should utilize both (along with pushing to be an active participant in any Section 504 considerations for their child)—but it’s not enough.
What if the “adverse reaction” is more than a low-grade fever. What if it’s sudden death—as was allegedly the case with 14-year-old Matthew Hohmann in 2004? As reported at the time at abcnews.go.com, his parents are convinced that Matthew’s Adderall XR is responsible for his death. And what of the FDA’s Medwatch reporting system then? No, what you need then is an attorney.
What if it’s Maryanne Godboldo of Michigan who’s fighting for her custody of her daughter following a drawn out battle with CPS (Child Protection Services) over her stopping her daughter’s use of the psychotropic drug, Risperdal?
And see, Godboldo’s story didn’t start there—it started when she and her daughter wanted to enter the public school system after several years of homeschooling—and the school required immunizations. Godboldo noticed uncharacteristic behavioral changes in her daughter within two weeks of the immunizations. And she began—as any responsible parent would—to try to seek help for her child.
Her suspicion that the immunizations might be the cause of the behavioral change was dismissed, and doctors prescribed psychotropic drug therapy for her daughter, which Godboldo later discontinued. The result? Health care providers claiming Godboldo medically abused her daughter, and CPS stepping in to remove the child from the home.
Godboldo is now facing criminal charges for allegedly firing a gun shot at police when they came to take her daughter away. If indeed she did take aim, who as a parent among us can not sense her frustration, her desperation and her lack of any other recourse to have her voice heard?
Ditto Jim Kaiser, who actually felt he had no other option than to kidnap his own son a few years back in order to keep his child from being forced to take Adderall (see video above). What you don’t see in the video report on Kaiser is what he shares on his website, MyNameisBen.org, where he shares that his child’s mother has been diagnosed with bipolar disorder and he also shares “words that she personally wrote” in her journal about her feelings and her ability (or lack thereof) to take care of her son. Reading this, if indeed true, provides a bit of backstory to Kaiser’s fight for his son and, as with Godboldo above, would make any parent understand what drove him to kidnap his own child to save him from being drugged.
And, with the statistics for ADHD diagnosis on the rise—according to the CDC (cdc.gov), parent-reported ADHD diagnosis rose twenty-two per cent between 2003 and 2007—this is not an issue that’s going away any time soon.
The problem with the lame “follow the process” process to documenting a child’s diagnosis (or misdiagnosis, as with ADHD can be the case) and treatment is that, as a parent, there is no real recourse as Godboldo and Kaiser have found out. Following the rules and trying to do the right thing nets nothing except an up-to-date file in some school office somewhere. And it nets a young man—truly no longer a child, though legally so—reaching out on a Facebook page in a plea for help and a desire for action, somehow, some way.
My advice to this young man is this: Continue to question. Continue to vocalize. Continue to fight. You will be heard. File the reports and the paperwork to CYA (cover your a**)—or advocate for those you love to do so. Follow the “drugging our kids” stories that are garnishing national attention, and don’t be afraid to speak with an attorney who specializes in civil rights—or, though I hope it doesn’t come to this as it would mean injury is involved, pharmaceutical litigation or medical malpractice (lest we forget little Rebecca Riley).
And for the rest of us, it’s time to take note of this young man’s plea and do something.
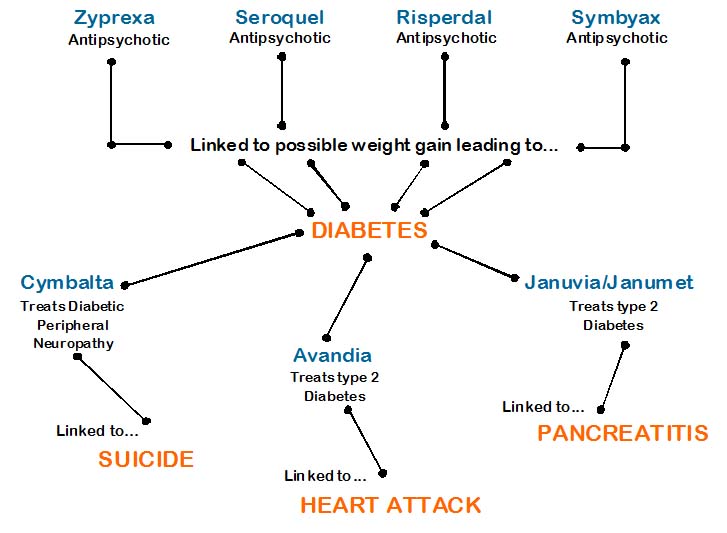
Kevin Bacon Game helps Decipher Diabetes-related Drug Lawsuits
September 2nd, 2010. By AbiK
If you’ve ever wondered why certain drugs (Zyprexa, Avandia, Seroquel, Cymbalta…to name a few) seem to have something in common, but you just can never really put your finger on it, well, it’s time to look to the Kevin Bacon Game (aka, Six Degrees of Separation, with some liberties taken) to find out what those connections are…so here goes…
Sex and Psych Drugs – Young Couples Beware
June 2nd, 2009. By Evelyn Pringle
Although the adverse effects of women taking psychiatric drugs while pregnant related to birth defects and infant withdrawal syndrome are often discussed or reported, the serious adverse effects on the sex lives and reproductive systems of millions of young couples are rarely mentioned.
Whatever the reason, due to the ever widening marketing campaigns by the psycho-pharmaceutical industry, young people need to be warned before they get conned into taking psychiatric drugs. Read the rest of this entry »
Lawsuits Needed Against Prescribing Quacks
April 24th, 2009. By Evelyn Pringle
On April 21, 2009, the Miami Herald reported that a 7-year-old boy in Florida, Gabriel Myers, had committed suicide by hanging himself with a detachable shower head in a bathroom of the foster care home he was placed in three weeks earlier. Read the rest of this entry »
Archive by Category
- Accidents (24)
- Airlines (9)
- Asbestos Mesothelioma (262)
- Automotive (25)
- Celebrity (14)
- Class Action (84)
- Complaints/Comments (15)
- Consumer Fraud (84)
- Contest (2)
- Court of Public Opinion (5)
- Crazy Sh*t Lawyers See (61)
- Criminal Law (4)
- Defective Products (111)
- DePuy ASR Hip Recall (2)
- Discrimination (22)
- Drugs/Medical (248)
- Elder Care Abuse (4)
- Emerging Issues (462)
- Employment (54)
- Environment (52)
- Financial (28)
- Food Illness (15)
- Human/Civil Rights (4)
- Insecurities (5)
- Insurance (16)
- Intellectual Property (16)
- Internet/E-commerce (19)
- lawsuits (161)
- Lawyers (20)
- Lawyers Giving Back (43)
- Lex Levity (10)
- Personal Injury (106)
- Pleading Ignorance (53)
- Real Estate (2)
- Recall (6)
- Scam (3)
- Securities (13)
- Settlement (81)
- Tort Reform (2)
- Totally Tortelicious (81)
- Veterans (11)
- Whistleblower (9)