Drugs/Medical Archive
Hey Guys, Are you in a State of Propecia Sexual Dysfunction?
June 10th, 2016. By AbiK
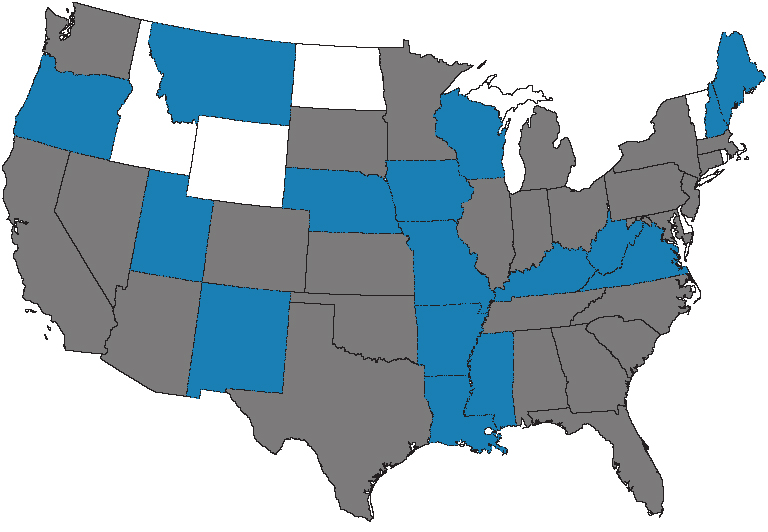
UPDATE: Since we first published this post (7/24/11), we reviewed the states in which we’ve received complaints regarding Propecia–some of which have had lawsuits filed. Forty-six US states have now been represented by Propecia complaints. The states shown in blue above are those that have seen complaints since the original post was published, below.
This whole Propecia sexual dysfunction thing got me wondering if there were any patterns (male pattern baldness aside) to where guys were experiencing the most negative Propecia sexual side effects, allegedly brought on by Propecia for hair loss treatment.
Being a data geek at heart, I took a look at where all these guys have been coming from—thinking that surely they’d hover around image-conscious L.A. or Miami—or in more major metro employment hotbeds where one might be concerned about age discrimination upon walking into an interview with a receding hairline—places like New York City or Boston or Chicago.
But it seems Propecia and its reported not-so-nice sexual side effects don’t discriminate—or at least not obviously so. Take a look above—the gray states are those from which LawyersandSettlements.com has received comments and complaints from guys sharing their Propecia sex problem stories. There’s no readily discernible pattern as to where guys who’ve taken Propecia and experienced sexual problems live. Propecia problems, it seems, have been happening allover. (And no, Hawaii and Alaska aren’t here, nor have I included Canada—but we’ve heard complaints from each).
To be clear, we’re not talking a low-key kind of sexual not-in-the-mood thing. These are guys who allege the mind is willing, wanting and very in the mood, but the body is not, shall we say, ramping up to support the mood. Alleged Propecia sexual side effects include a wide range of sexual dysfunction: erectile dysfunction, inability to ejaculate, low sperm count—leading to inability to conceive, and yes, even lack of sexual desire. Not the things a virile young (or older) man wants to be dealing with as he’s in the prime of his dating years or trying to start a family or just trying to remain intimate with his wife.
And, I don’t think I need to tell you that the situation doesn’t only affect the guys here—there’s that “other half” who’s involved. After a while, a Propecia victim may find himself absent-mindedly humming a Doors’ medley that started nicely enough with “Light my Fire” (as in, “C’mon baby…”) and ended with the more frustrating “Don’t you love her as she’s walking out the door”…as for some women, walking out will be exactly what they do.
My fear is that the above map is only the beginning as more men begin to come forward and share their stories. It takes guts to admit one’s short-comings and to reveal sexual inadequacy vs. dancing around it or remaining in denial about it takes a set of you know what—particularly when it’s as a result of trying to overcome another ego-deflator: hair loss. But if you’re in a state of sexual dysfunction and you think it’s a result of Propecia side effects, better to be in a state that’s complaining vs. a state of denial. Get some help.
Testosterone: Mojo for Hire or in Bed with a Liar?
April 21st, 2014. By Hunter West
 Funny thing happened on the way to the fountain of youth: the road was littered with lotions and potions, Botox and fillers, false claims and false tits—and, of course, facelifts. Even vaginal rejuvenation. Everything a gal could want for trying to hold onto what nature will ultimately take away—regardless of whether a false veneer of youth is gotten and paid for, or not. But left in dust behind every woman of the desperately-clutching-to-twenty-nine set was someone who did not have such readily available magic…such opportunity: the common, and aging, man (well, Bruce Jenner and David Hasselhoff plastic surgery rumors aside.)
Funny thing happened on the way to the fountain of youth: the road was littered with lotions and potions, Botox and fillers, false claims and false tits—and, of course, facelifts. Even vaginal rejuvenation. Everything a gal could want for trying to hold onto what nature will ultimately take away—regardless of whether a false veneer of youth is gotten and paid for, or not. But left in dust behind every woman of the desperately-clutching-to-twenty-nine set was someone who did not have such readily available magic…such opportunity: the common, and aging, man (well, Bruce Jenner and David Hasselhoff plastic surgery rumors aside.)
Ah, but the conundrum was worse than imagined. Never mind that men may have felt excluded from the pulled face and tummy tucked echelons of society. Another quandary was rearing its ugly head, tucked away in the offices, cubes and conference rooms of Big Pharma: the very product that could tout itself as the male fountain of youth had only limited reach.
The product? Testosterone.
Testosterone: Who Needs you Baby?
Ok, there’s little doubt that the current debate over low-testosterone has given rise to the broader issue of need vs. want. Medical necessity vs. vanity.
Is this something I truly need, or am I just being sold a bill of goods…nothing more than a well-marketed lie?
The pharmaceutical industry (aka Big Pharma) takes up a huge footprint in American commerce. Pharmaceuticals have helped us live longer, free of disease. Ongoing research—and the treatments that follow—has succeeded in elevating our lives and collective life expectancy well beyond the point at which our forefathers checked out for good.
But where will all this end? And will Big Pharma remain unsatisfied until they have a pill for every small, niggling thing that emerges as the natural process of aging?
Need to get up several times at night to go to the bathroom? There’s a pill for that. Wrinkles? There’s Botox for that. A middle-aged man with flagging energy? There’s a testosterone topical gel for that…
It seems—at least through the lens of the pharma co’s—the fountain of youth can, and should, be found at your friendly, neighborhood drug store.
On the testosterone front, there is fierce debate. And with good reason. Testosterone supplementation, originally approved for conditions such as hypogonadism (undescended testes) and extremely low levels of testosterone (or no testosterone at all), is and has been for decades a legitimate treatment for men suffering from low testosterone.
Like any and every drug on the market, there are testosterone side effects. Testosterone can spur the production of red blood cells, and in some cases a level of testosterone that is too high can lead to cardiovascular issues such as testosterone stroke or testosterone heart attack. But drugs, medical devices and treatments are approved by the US Food and Drug Administration (FDA) based on the mantra that so long as a drug’s benefits outweigh the potential risks for the intended constituency of patients, then the drug is appropriate for the market.
Manufacturers are always lauding their drugs as ‘safe and effective.’ But the FDA talks only in terms of ratios involving benefit v. risk. The risk of side effects is an unspoken given.
Thus, testosterone. For patients with hypogonadism or otherwise extremely low levels of testosterone, the benefits of supplementation to elevate quality of life can be profound, and well worth the risks.
Testosterone supplementation does have its place. But should it be used to treat the natural symptoms of simply getting older?
Low-T: It’s a Thing!
Most people not associated with pharmaceutical companies that stand to make huge profits through the identification of new markets, would probably agree that it should not. But that doesn’t stop an already-wealthy industry from the need and greed to be richer still, by way of a limitless quest for new markets in order to flog products well beyond the uses for which they were approved in the first place.
You don’t have to stretch too far to imagine that some marketing whizzes in a Big Pharma back room—their own testosterone racing—sniffed around for new testosterone markets and came upon a Venn diagram showing the sweet spot where age-induced Low-T intersects with an ever-growing baby boomer population. The solution! There it was! The Low-T Low Hanging Fruit! Yes, indeed there was a broader market to be tapped that could deliver broader market saturation: the aging boomer whose energy is flagging, breasts are sagging (probably caused by another drug), overweight, a bit lethargic, and not quite as prolific in the bedroom as they used to be. Tough to get to the arena for that late-night pickup hockey game, when you’re falling asleep after dinner…The storyboards were already starting to fly off the pads of the wannabe Art Directors!
But what’s even better than identifying that hypothetical sweet spot in a marketing plan? Finding the reality that…men actually CARE about this stuff now. Suddenly they’re not satisfied with slowing down at middle age. Self-improvement suddenly matters, and the pursuit of the Fountain of Youth is no longer reserved for women.
That’s right. Let’s tell the men we have their back, with products that can boost their energy, help them lose weight, help them think more clearly, and return them to hero status in the bedroom.
Next thing you know ‘Low-T’ (as marketing catchphrase) was born. And now Low-T has become ‘A Thing’.
Thing that Makes you go Hmm?
A testosterone lawsuit recently filed by a plaintiff who succumbed to flashy marketing and at 61 was hospitalized with blood clots, zeroes in on the origins of the Low-T phenomenon, insights gleaned from a revealing expose appearing in The New York Times last fall.
‘Selling That New Man Feeling’ (New York Times, 11/23/13) paints a vivid picture of just how we got here—and how plaintiff Roger Gibby found himself in the hospital with blood clots in his legs and lungs.
“None of the testosterone products have indications for weight loss, increasing energy or improving mood,” said Andrea Fischer, a spokeswoman for the FDA, in comments published in The New York Times. In other words, testosterone products were never intended for anything other than hypogonadism and extremely low levels of testosterone that required medical intervention.
Author of Low-T Quiz: it’s a “Crappy Questionnaire”
And yet this incredible market has risen like a Phoenix—with annual sales in the billions of dollars—built on a foundation of male vanity, intensive advertising, dubious need and a $40,000 questionnaire jotted down within 20 minutes on toilet paper during a bathroom break.
Say what?
Dr. John E. Morley, director of endocrinology and geriatrics at St. Louis University School of Medicine who has previous ties to the pharmaceutical industry through paid speaking engagements, was asked by Organon BioSciences, a Dutch pharmaceutical company, to come up with a screening questionnaire that highlighted symptoms common to older men with low testosterone. He was told to keep it short and make it somewhat sexy.
In exchange for 20 minutes in the bathroom, Morley’s employer received a $40,000 research grant. And those 20 minutes in the bathroom has gone on to become an online staple now known as the ‘Is It Low-T Quiz.’
Dr. Morley admitted to The New York Times that it wasn’t his best work. ‘I have no trouble calling it a crappy questionnaire,” Morley told The Times. “It is not ideal.”
That quiz is now utilized by many of the biggest manufacturers of testosterone supplements. The idea is to attempt a self-diagnosis of ‘Low-T’ online, then bring that to your doctor to lobby for testosterone supplementation. And many doctors have bought into the idea of testosterone as a viable method for restoring sagging energy levels.
That’s not what testosterone supplements were approved for. But then, doctors have the legal, moral and ethical authority to circumvent FDA approval parameters and prescribe products for use off-label.
Here’s the question: have doctors bought into the hype, in the same fashion as their sagging, flagging male patients searching for that fountain of youth?
Who’s Buying it?
There is little doubt that manufacturers of testosterone supplements identified a market and exploit it for all its worth. In its statement of position, the Gibby Low-T lawsuit picked up on the naming of two AbbVie executives by Medical Marketing & Media as “the all-star large pharma marketing team of the year,” for 2013. The New York Times noted that the plaudits were for promotions involving AndroGel and unbranded efforts to advance Low-T.
“It didn’t hurt that baby boomers have proven less than shy about availing themselves of any product that they believe will increase their quality of life,” an article in the magazine said. AbbVie’s unbranded site DriveForFive.com, was lauded for encouraging men to have regular checkups and to ask their doctors about five tests, among them checks for cholesterol, blood pressure—and testosterone.
AbbVie takes the high road, officially stating that all it’s doing is making men aware of the potential for low testosterone, the potential benefits of testosterone supplementation, and the importance of starting a dialogue with their doctor on the issue.
But even the availability of the on-line screening quiz, dubbed ADAM, flies in the face of a position maintained by The Endocrine Society, which recommends against screening of the general population for Low-T. The Society makes the point that testosterone levels in most men fluctuate a great deal depending on several factors, from sleep levels to stress, or time proximity to the latest meal consumed. Even the time of day, or if a favorite sports team is losing, can affect testosterone levels.
As such, a single testosterone reading may not indicate a problem. But that hasn’t stopped Big Pharma from advancing Low-T to status of ‘A Thing’ that brings in billions of dollars each year. And one study says that men have bought into it so conclusively, a full 25 percent of men who were given testosterone prescriptions did not have the requisite blood work to back that need up.
Hence, the suggestion that doctors have bought into it, too.
This has fueled the debate between those who believe testosterone is not the pariah many make it out to be, vs. the anti-testosterone camp who want supplements banned.
Both sides have points. For some men, testosterone supplementation is both necessary and a godsend. And as has been historically the case prior to the recent creation of the ‘Low-T’ phenomenon, testosterone supplementation as a response to a real medical condition such as hypogonadism, or extremely low levels of testosterone based on sound medical testing and diagnosis, is indeed appropriate.
Just as there are risks associate with advanced levels of testosterone, so too are there risks associated with seriously low levels.
However, helping to fuel the debate over testosterone side effects on the too-high side, is the proliferation of the Low-T market that suggests there are a lot of men out there having been persuaded to adopt testosterone supplementation in the absence of sound, medical need.
And that puts them at risk for testosterone stroke, or testosterone heart attack, needlessly—perhaps all because Big Pharma saw a market with potential dollar signs, and went to work…
Proponents of testosterone as a means to foster men’s health have found fault with some studies—and one in particular—that paint testosterone supplementation as a villain.
There have also been accusations of fear-mongering levied against the media and anti-testosterone advocates, who are accused of needlessly inflating worry and blowing the issue out of proportion.
In contrast, there are those who accuse the testosterone industry and pro-testosterone advocates of ‘disease-mongering,’ by promoting a need—a problem—that doesn’t really exist for the general population.
In so doing, testosterone is flying back and forth unabated, as tensions continue to rise amid a debate that tries to differentiate between the natural processes of aging vs. the wisdom to intervene needlessly in such a natural process.
Medical need is one thing. Exploiting a potential market is quite another.
What is the need, really? And do we put our faith in an industry, with already deep pockets and looking to make them deeper, that tries to convince us that we need this stuff?
I don’t think so. I’ll take the unbiased, unvarnished opinion of my doctor who stands to reap no reward other than the realization of a healthy patient who thinks it’s okay to grow old gracefully. But I’ll be damned if I’ll give money to a pharmaceutical company claiming to have my best interest at heart, because they don’t. They have a conflict. If they convince me I need testosterone, they make a buck.
A long time ago I was taking a business course, and the course lead asked of the room, the reason why people go into business. To fulfill a dream? To help your fellow man? To make the world a better place? To leave behind a legacy?
Nope. All wrong answers.
The reason—is to make money. Profit. That’s it. Everything else is secondary.
It’s enough to get my testosterone racing—what little I have left…
Law News For You: Reality TVM – Mesh Implant Victims Speak Out
June 5th, 2013. By AbiK
Infographic: Depakote Birth Defects, Off-Label Marketing by the Numbers
March 1st, 2013. By AbiK
Recently, a small British study that was published in the Journal of Neurology, Neurosurgery & Psychiatry (1/30/13), suggested women who take valproate while pregnant have an increased risk of having children with autism and neurodevelopment problems. The study also suggested that damage to the fetus happens during the early stages of pregnancy.
Valproate-based anti-convulsants (drugs used to treat seizure disorders including epilepsy) include brand-name drugs Depakote, Depakene, Depacon and Stavzor–all of which are pregnancy risk Category D drugs. Category D drugs have shown positive evidence of fetal risk based on adverse reaction data from investigation or marketing experience or clinical studies in humans.
The Depakote infographic below highlights some of the potential adverse effects in newborns that have been associated with taking Depakote during the first trimester of pregnancy.
Stryker Hip Recall, Broadspire & the Meaning of Two Words
February 7th, 2013. By AbiK
 Not since the days of Monica Lewinsky has parsing the meaning of a word—or two in this case—been worthy of such scrutiny. The words, “if necessary” in this “Claims & Reimbursement FAQs” link on the Stryker website should perhaps make any hip replacement patient who’s been affected by the Stryker Rejuvenate and ABG II modular-neck hip stems recall suspect.
Not since the days of Monica Lewinsky has parsing the meaning of a word—or two in this case—been worthy of such scrutiny. The words, “if necessary” in this “Claims & Reimbursement FAQs” link on the Stryker website should perhaps make any hip replacement patient who’s been affected by the Stryker Rejuvenate and ABG II modular-neck hip stems recall suspect.
Why?
Well, a little walk down memory lane on LawyersandSettlements.com brings up a story we had published back when Broadspire was the third-party claims administrator brought in by J&J to handle DePuy hip replacement claims.
At the time, a Reuters article noted that the move means that Broadspire’s physicians—not the patient’s physician—will determine whether a hip should be replaced. This means that even if the patient’s physician recommends replacing the hip, if a Broadspire physician disagrees with the decision, Johnson & Johnson may not pay to cover the costs of that surgery.
So is history repeating itself?
Clearly by inserting the words “if necessary” in discussing defective hip replacement revision surgery reimbursements Broadspire retains the right to deem whether a revision surgery was necessary, or not; the “not” undoubtedly necessitating a denial—or reduction—of reimbursement.
What we’re hearing from attorneys like Ben Stewart of Stewart Law Group PLLC., who’s familiar with defective hip lawsuits, is that before a patient submits any paperwork with Broadspire—or signs anything—for a Stryker hip reimbursement claim, he or she should first talk to a lawyer.
Archive by Category
- Accidents (24)
- Airlines (9)
- Asbestos Mesothelioma (262)
- Automotive (25)
- Celebrity (14)
- Class Action (84)
- Complaints/Comments (15)
- Consumer Fraud (84)
- Contest (2)
- Court of Public Opinion (5)
- Crazy Sh*t Lawyers See (61)
- Criminal Law (4)
- Defective Products (111)
- DePuy ASR Hip Recall (2)
- Discrimination (22)
- Drugs/Medical (248)
- Elder Care Abuse (4)
- Emerging Issues (462)
- Employment (54)
- Environment (52)
- Financial (28)
- Food Illness (15)
- Human/Civil Rights (4)
- Insecurities (5)
- Insurance (16)
- Intellectual Property (16)
- Internet/E-commerce (19)
- lawsuits (161)
- Lawyers (20)
- Lawyers Giving Back (43)
- Lex Levity (10)
- Personal Injury (106)
- Pleading Ignorance (53)
- Real Estate (2)
- Recall (6)
- Scam (3)
- Securities (13)
- Settlement (81)
- Tort Reform (2)
- Totally Tortelicious (81)
- Veterans (11)
- Whistleblower (9)
