LAWSUITS NEWS & LEGAL INFORMATION
Mirena IUD Pseudotumor Cerebri and Uterine Perforation Lawsuits
Read our Mirena IUD FAQ
Were you looking for FDA Receives Hundreds of Complaints About Essure Contraceptive Coils lawsuits?
By Jane Mundy
Mirena pseudotumor cerebri lawsuits allege that Bayer, the company that makes Mirena IUD, did not warn consumers and the medical community of pseudotumor (PTC) risks—also known as intracranial hypertension (IIH)-- and other serious Mirena side effects such as mirena uterine perforation. From 2000 to 2013 the FDA received more than 70,000 reports of Mirena IUD side effects, and hundreds of Mirena intracranial hypertension lawsuits claim that Bayer withheld information from consumers about the importance of seeking immediate medical attention if they experience severe headaches, vision problems or other symptoms of PTC after Mirena is implanted.
En Español MIRENA DUI
FREE MIRENA IUD LAWSUIT EVALUATION
Send your Mirena IUD claim to a lawyer who will review your claim at NO COST or obligation.
GET LEGAL HELP NOW
GET LEGAL HELP NOW
Mirena IUD Side Effect Claims Being Investigated
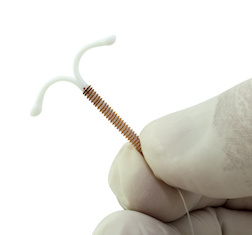 Mirena was approved by the FDA in 2000 as a contraceptive device and in 2009 it was approved for the treatment of heavy menstrual bleeding for women who prefer an IUD as their method of birth control. The Mirena IUD is a T-shaped device that is implanted directly into the uterus and can remain in place for up to five years. It works by slowly releasing levonorgestrel, a synthetic progestogen, to prevent pregnancy and to treat heavy menstrual bleeding. (Progestogen is a naturally occurring hormone.) Levonorgestrel is commonly used in combined oral contraceptives, and the intrauterine form of levonorgestrel (IUL) is marketed as Mirena® in North America.
Mirena was approved by the FDA in 2000 as a contraceptive device and in 2009 it was approved for the treatment of heavy menstrual bleeding for women who prefer an IUD as their method of birth control. The Mirena IUD is a T-shaped device that is implanted directly into the uterus and can remain in place for up to five years. It works by slowly releasing levonorgestrel, a synthetic progestogen, to prevent pregnancy and to treat heavy menstrual bleeding. (Progestogen is a naturally occurring hormone.) Levonorgestrel is commonly used in combined oral contraceptives, and the intrauterine form of levonorgestrel (IUL) is marketed as Mirena® in North America.Mirena lawyers are reviewing claims for women nationwide and in Canada who have experienced side effects of Mirena, such as:
- Idiopathic Intracranial Hypertension (IIH);
- Pseudotumor Cerebri (PTC);
- Migration or Perforation Injury Resulting in Surgical Removal Unintended Pregnancy Resulting in Birth Defects or Injury to Mother;
Mirena Pseudotumor Cerebri Lawsuits
According to more than 100 Mirena PTC and IIH lawsuits, Bayer sold the IUD birth control without providing adequate warnings and information about the risk of these serious complications. They claim that Bayer withheld information from consumers and the medical community about the importance of seeking immediate medical attention if they experience severe headaches, vision problems or other symptoms of PTC after Mirena is implanted.
The Mirena prescribing information says that the IUD should be used with caution in patients who have “migraine, focal migraine with asymmetrical visual loss or other symptoms indicating transient cerebral ischemia.” This information could mean the possibility of an ischemic stroke, which involves a blood clot in the brain, but Mirena does not warn anywhere about the possibility of idiopathic intracranial hypertension or pseudotumor cerebri.
In April, 2017 the IIC and PTC lawsuits were centralized in a Multi-District Litigation (MDL). The U.S. Judicial Panel on Multidistrict Litigation (JPML) established coordinated pretrial proceedings for all federal complaints pending in the MDL. The U.S. District Judge presiding over these federal cases has approved filing of new cases directly in the multidistrict litigation (MDL).
By April 26, over 100 federal Mirena lawsuits have been consolidated (MDL No. 2767) in the District of New York.
In January, 2017 a Georgia woman filed a complaint against Bayer claiming Mirena caused a dangerous buildup of fluid pressure around her brain, known as idiopathic intracranial hypertension (IIH) or pseudotumor cerebri (PTC), resulting in severe vision problems and nerve damage to her eye muscles. Maria Pitts filed her complaint in the in the U.S. District Court for the District of New Jersey, alleging that Bayer failed to warn about the risk of IIH/PTC complications from Mirena.
In April 2016 a Mirena PTC lawsuit was filed by a Maryland woman alleging the birth control device caused her to develop idiopathic intracranial hypertension (IIH). Daphne Houck claims she experienced these symptoms shortly after the Mirena was implanted. She followed up with an MRI and CT scan that diagnosed pseudotumor cerebri and underwent a lumber puncture to remove excess cerebrospinal fluid from the skull.
In September 2015 a Florida woman filed a complaint in the U.S. District Court for the Western District of Tennessee, alleging that Mirena caused her to suffer severe migraines, cognitive problems, vision problems, memory loss and other problems. Nicole Hamill claims that she experienced PTC symptoms just one month after being implanted with the IUD in 2004. Hamill required lumbar punctures to relieve intracranial pressure and had the Mirena IUD removed. However, the punctures do not cure the condition. In March 2014 the first Mirena pseudotumor cerebri(PTC) and/or idiopathic intracranial hypertension (IIH)lawsuit was filed in Alabama. A federal judge allowed the lawsuit to proceed after plaintiff Shenika Houston sufficiently claimed negligent design, strict liability, breach of express warranty, and fraud. The lawsuit states that patients who develop PTC might continue to use Mirena because the label does not include warnings about this potential risk. This could contribute to the development or progression of PTC or IIH.
In May 2014, Law 360 reported that lawyers, on behalf of plaintiffs, asked a panel of federal judges to centralize nine Mirena lawsuits involving PTC/IIH into an MDL in the U.S. District Court for the Middle District of Tennessee, which is separate from the MDL 2434 (above).
Plaintiffs allege that Bayer failed to adequately warn about the link between the hormone levonorgestrel, present in Mirena, and PTC/IIH. The lawsuits allege that levonorgestrel can elevate levels of cerebrospinal fluid in the brain, which increases pressure in the brain, similar to a tumor.
All plaintiffs claim that Bayer knew or should have known about the link between Mirena and levonorgestrel and PTC, and Bayer should have provided stronger warnings about the risk of PTC/IIH.
The intracranial hypertension from Mirena often presents as a swelling of the optic nerves, called papilledema. Bayer has included information about the risk of papilledema on its warning labels in several countries, such as South Africa and Hong Kong. It has not given doctors or Mirena consumers in the US the same warning.
Background: Mirena and Pseudotumor Cerebri/Idiopathic Intracranial Hypertension
In Mirena clinical trials, 7.7 percent of women reported headaches/migraines.
The Mayo Clinic lists the following symptoms associated with PTC:
- moderate to severe headaches originating behind the eyes that worsen with eye movement;
- ringing in the ears that pulses in time with a person' heartbeat;
- dizziness, nausea or vomiting;
- blurred vision and/or double vision;
- seeing light flashes;
- temporary or passing blindness;
- neck, shoulder or back pain.
Mirena Pseudotumor Cerebri Studies
In April 2017, the journal Neuro-Ophthalmology published “An Estimation of the Risk of Pseudotumor Cerebri among Users of the Levonorgestrel Intrauterine Device.” After reviewing medical records from databases, researchers found that Mirena users were over seven times more likely to develop pseudotumor cerebri. This study suggests that there is compelling evidence of an association between the IUD and brain injuries.
Mirena Uterine Perforation
For claims involving injuries caused by migration of Mirena and perforation of the uterus, a Multi-District Litigation (MDL 2434) has been centralized in federal court in New York, involving more than 500 lawsuits. Nearly 800 additional lawsuits are pending in state court in New Jersey.
Mirena IUD Legal Help
If you or a loved one has suffered illness or an adverse health event resulting from the use of this product, please click the link below and your complaint will be sent to a lawyer who may evaluate your claim at no cost or obligation.
Last updated on
MIRENA IUD LEGAL ARTICLES AND INTERVIEWS
“I want this Mirena IUD out, over and done with.”
Toronto, ON. Nickey followed her doctor’s orders, and the Mirena manufacturer’s orders: five years after the IUD was implanted, she made an appointment to have it removed. But it wasn’t that easy. Next month will be the fourth, and she hopes final, attempt to have all of the birth control device removed from her uterus.
“On the first attempt, my OB/gyn couldn’t find the string so he sent me to another specialist where I had an ultrasound to see where exactly this Mirena was,” says Nickey. “He found the Mirena in my uterus—where it was supposed to be—but when he tried to remove it, the metal piece broke.”
Next up, Nickey had to undergo a general anesthetic, but her doctor told her during the follow-up appointment that the Mirena had perforated her uterus. “He told me that a Da Vinci surgical robot was supposed to have been used in this procedure but it wasn’t available that day,” Nickey says. he didn’t tell Nickey when and how her uterus was perforated by the Mirena. “He is definitely going to use the robot next month to retrieve this broken piece— it will be the fourth attempt to have this Mirena entirely removed.”
A study published by the International Journal of Women’s Health concluded that IUD perforation, “is a rare but serious complication of IUD use”. If a device perforates the uterine wall, it can continue to migrate elsewhere in the body and extensive surgery may be necessary to repair the damage. Nickey is 33 years old and the mother of a 10-year-old boy. She wants to have another child, so this complication is extremely worrisome.
Given the number of lawsuits filed against the manufacturer, Bayer Healthcare, alleging permanent injuries and trauma due to complications that necessitated surgical removal, IUD perforation is not so rare. (The study was conducted in 2014). Between 2000 and 2013, the FDA had received 70,072 complaints about the Mirena IUD, according to a report by
7 Action News investigators. (Here is the FDA’s adverse event reporting system, called FAERS.)
“Since the first surgery to have the Mirena removed I have had such heavy bleeding that I needed to take iron pills and often found myself shaking,” says Nickey. “As well, I had a lot of pain after the surgery—I’m not looking forward to the surgery on January 24th. And it’s a one-hour drive to the hospital. Who is paying for my time?
“If I knew what I know now about Mirena, I would never have gotten this IUD. I’m weighing my options about other kinds of birth control but before that, I have to get my body back to normal. And I am concerned that I have damage to my uterus because I want more kids one day. My doctor knows I want more kids and he has given me options if it cannot be removed. If push comes to shove, he mentioned hysterectomy. Emotionally and physically, this IUD has taken a lot out of me…I just want it out.”
If the Mirena has perforated the uterine wall, robotic surgery or laparoscopic surgery is necessary. A number of women who have undergone surgery to have the device removed have charged Bayer with manufacturing a defective product. They seek damages to cover medical expenses, lost wages, and ongoing care needs, and to compensate them for pain and suffering. Sadly, many women have also undergone hysterectomies.
READ MORE
Lawsuit Alleges Hormone Coated Mirena IUD Can Lead to Blindness

Mirena IUD Lawsuit Alleges Grievous Mirena IUD Side Effects


December 30, 2017
Toronto, ON. Nickey followed her doctor’s orders, and the Mirena manufacturer’s orders: five years after the IUD was implanted, she made an appointment to have it removed. But it wasn’t that easy. Next month will be the fourth, and she hopes final, attempt to have all of the birth control device removed from her uterus.
“On the first attempt, my OB/gyn couldn’t find the string so he sent me to another specialist where I had an ultrasound to see where exactly this Mirena was,” says Nickey. “He found the Mirena in my uterus—where it was supposed to be—but when he tried to remove it, the metal piece broke.”
Next up, Nickey had to undergo a general anesthetic, but her doctor told her during the follow-up appointment that the Mirena had perforated her uterus. “He told me that a Da Vinci surgical robot was supposed to have been used in this procedure but it wasn’t available that day,” Nickey says. he didn’t tell Nickey when and how her uterus was perforated by the Mirena. “He is definitely going to use the robot next month to retrieve this broken piece— it will be the fourth attempt to have this Mirena entirely removed.”
A study published by the International Journal of Women’s Health concluded that IUD perforation, “is a rare but serious complication of IUD use”. If a device perforates the uterine wall, it can continue to migrate elsewhere in the body and extensive surgery may be necessary to repair the damage. Nickey is 33 years old and the mother of a 10-year-old boy. She wants to have another child, so this complication is extremely worrisome.
Given the number of lawsuits filed against the manufacturer, Bayer Healthcare, alleging permanent injuries and trauma due to complications that necessitated surgical removal, IUD perforation is not so rare. (The study was conducted in 2014). Between 2000 and 2013, the FDA had received 70,072 complaints about the Mirena IUD, according to a report by
“Since the first surgery to have the Mirena removed I have had such heavy bleeding that I needed to take iron pills and often found myself shaking,” says Nickey. “As well, I had a lot of pain after the surgery—I’m not looking forward to the surgery on January 24th. And it’s a one-hour drive to the hospital. Who is paying for my time?
“If I knew what I know now about Mirena, I would never have gotten this IUD. I’m weighing my options about other kinds of birth control but before that, I have to get my body back to normal. And I am concerned that I have damage to my uterus because I want more kids one day. My doctor knows I want more kids and he has given me options if it cannot be removed. If push comes to shove, he mentioned hysterectomy. Emotionally and physically, this IUD has taken a lot out of me…I just want it out.”
If the Mirena has perforated the uterine wall, robotic surgery or laparoscopic surgery is necessary. A number of women who have undergone surgery to have the device removed have charged Bayer with manufacturing a defective product. They seek damages to cover medical expenses, lost wages, and ongoing care needs, and to compensate them for pain and suffering. Sadly, many women have also undergone hysterectomies.
READ MORE
Lawsuit Alleges Hormone Coated Mirena IUD Can Lead to Blindness

November 9, 2017
Oakland, CA: The next twelve months will be critical for Mirena IUS (Intrauterine System) contraceptive plaintiffs currently engaged in a Multi-District Litigation lawsuit against Bayer alleging the hormone coated device leads to an intracranial condition with the potential to cause permanent blindness in some users. READ MORE
Mirena IUD Lawsuit Alleges Grievous Mirena IUD Side Effects

November 5, 2017
New York, NY: A Mirena side effects lawsuit filed late last month accuses the manufacturer of the popular IUD of not doing enough to warn consumers about the potential for pseudotumor cerebri, or PTC, associated with the long-term birth control device. According to Court records the plaintiff has suffered from vision problems and a build-up of fluid since acquiring the device. READ MORE
READ MORE Birth Control Settlements and Legal News
READ MORE Drugs/Medical Settlements and Legal News
READ MORE Personal Injury Settlements and Legal News
READ MORE Drugs/Medical Settlements and Legal News
READ MORE Personal Injury Settlements and Legal News

READER COMMENTS
Michelle Hong
on
Koutdoors
on
Kait Peabody
on
Rebecca darcy
on
ALIYA DIAZ
on
Dorian Estes
on
Trina
on
If I had knew before this I would have never gotten this and sadden that none of the medical professionals said anything after I told them about my painful breast and blanking out when trying to recall things.
Erika Byrd
on
Mimi
on
suzette calitz
on
Stephanie davis
on
Florida girl
on
Theresa west
on
Abby Fox
on
Heather white
on
There's another issue you haven't addressed
on
Kristin
on
Louisiana
on
New Mexico
on
Anonymous
on
I initially had the merina implant to regulate my periods. I had wanted the mirena out the very next day after the Dr. put it in. My body just felt weird. I ended up having nausea, headaches, foggy thinking, extreme fatigue, constipation, abdominal bloating, severve moodiness, depression urinary tract infections, severe abdominal pain. So I went to the Dr. to talk about how I was feeling and my concern that I felt the mirena had moved .
He said it was in place and he refused to have it remove it and said give it time.
I went to ER to get it out but they wouldn't either. So against my better judgment I complied with their direction.
Meanwhile my symptoms got worse . The bleeding continued to get heavier and I ended up bleeding ever since the insertion
on September 19,2013 until April 23, 2014.
I went to the ER numerous times with a diagnosis of dysfunction uterine hemorrhage. On Saturday October 19,2013 I had a lot of abdominal pain very similar to contractions during childbirth .Huge clots kept coming out. I had never seen such large blood clots ranging from the smallest about the size of a golf balls to the size of a softballs. In one of the blood clots was the merina. I put the merina in a in a zip lock bag along with some blood clots and went to the emergency room. The zip lock bag was almost filed to the top with clots. By taking the zip lock bag to the emergency room I was hoping they would send it to the pathology lab in hopes of finding out why I was bleeding so much. I left without any more information than I had before I
went there.
I ended up having three blood transfusions in a six month period. I also had to face the fact that my hair was falling out alot. I ended up spending alot of time in the shower.
I finally stopped going to the Dr. in December because he was unable to stop the bleeding, despite giving me bith control pills, the Depo-Provera shot repeatedly as well as the ER trying with various medication with no success.
At that point I gave up and was just waiting to bleed to death. My hemorrhaging was so bad that I rarely left the house. Eventually I found another Dr. and she ended up giving me D and C, which stopped the bleeding on April 23, 2014.
The implant is out however, I still am dealing with the aftermath. My Dr. said if it starts up again, we may need to explore the option of a hysterectomy. The teacher I have worked with for years had to get another educational assistant because I was unable to work. I was not working for the entire year which meant I was unable to provide for my family. My life was put on hold because I thought I was going to bleed to death. My physical health still is not back to what is before this happened. My body is still trying to recover after this horrific ordeal. I'm still have a lot of emotional scars from the trauma, I continue to experience on and off again pain in my lower abdominal as well as lower back pain, which I never had before the implant. My sex life has changed, I continue to fell pain while having sex. I just want to get my life back to how it was before the implant. At this time I need some kind of resolution in order to put this nightmare behind me.
Please note:
The implant was not surgically removed.
Kansas
on
Mississippi
on
Nevada
on
British Columbia
on
California
on
Ontario
on
Kansas
on
Texas
on
Washington
on
Utah
on
Ontario
on
Florida
on
Anonymous
on
British Columbia
on
Arkansas
on
Ontario
on
-cyst growing out of the uterus wall
-bloating and pain always in the lower part of the stomach and also in the uterus
California
on
Michigan
on
damages suffered...loss of income endless medical bills and extreme pain and suffering
Pennsylvania
on
California
on
Florida
on
Indiana
on
North Carolina
on
Arkansas
on