LAWSUITS NEWS & LEGAL INFORMATION
Birth Injury & Birth Defect Lawsuits
Were you looking for Birth Defect and Birth Injury lawsuits?
By Heidi Turner
Birth injuries and birth defects are injuries that an infant is born with, either due to harm caused while in the womb or injuries sustained during birth. Numerous drugs have been associated with different types of birth defects, with varying degrees of severity. In some cases, the birth defects cause permanent injury or disability, in others the defect can be rectified through surgery. All cases, however, cause stress for the family and the infant. Birth defects lawsuits have been filed against the people or organizations whose actions may have resulted in infant defects.
Infants who are born with difficulties in how their organs or body parts are formed or work have birth defects. According to the National Institutes of Health, these defects are broken down into structural defects (part of the body is missing or malformed), and functional or development birth defects (including problems with the nervous system, the brain, or the senses). Some birth defects can be fixed with surgery or other interventions, while other defects are permanent.
Birth defects can be caused by a range of factors, including genetic problems or chromosome problems. But in some cases, infants are exposed to medications, chemicals or other toxins while in the womb that can affect their development. Meanwhile, injuries sustained during birth may also cause lifelong difficulties.
In cases where negligence, malpractice, or other unethical activity has occurred and that activity has resulted in birth defects, lawsuits can be filed against the people or organizations involved.
A variety of medications have been linked to different birth defects. In some cases, studies have found conflicting evidence regarding the link between medications and birth defects. This is partially because it is unethical for researchers to test medications on pregnant women.
As of 2015, the FDA changed how drugs are labeled in relation to pregnancy. Under the new guidelines, drug labels will include three subsections—"Pregnancy," "Lactation," and "Females and Males of Reproductive Potential." Those subsections will include a summary of the risks of using a drug during pregnancy or lactation and information that will help medical professionals make prescribing decisions. This new rule replaces the letter categories, which were previously used to classify medications.
In the past, the FDA classified drugs in five pregnancy categories (this system has since been replaced but is still valid as drugs already on the market are slowly moved to the new classification):
Selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs) are medications used to treat patients with depression. The medications have been linked to a variety of birth defects including congenital (heart) malformations when women took SSRIs during different stages of pregnancy. Although studies have reached conflicting conclusions regarding the potential link between SSRIs/SNRIs and birth defects, lawsuits have been filed against the makers of some SSRIs/SNRIs.
Some birth defects reportedly linked to SSRIs/SNRIs include persistent pulmonary hypertension of the newborn (PPHN), a disorder of the lungs in which the arteries are restricted, causing blood pressure in the pulmonary artery to drastically increase. PPHN can cause shock, heart failure, seizures, organ damage, and death.
Included among the SSRI and SNRI drugs potentially linked to birth defects are:
SSRIs SNRIs
Lyrica (known generically as pregabalin) is a pain medication used to treat nerve pain linked to fibromyalgia, shingles, and diabetes. Studies suggest women who take Lyrica during their first trimester are at an increased risk of having a baby with skeletal or neural tube defects.
Zofran is an anti-nausea medication often prescribed off-label to treat women suffering from severe morning sickness. It has been linked to an increased risk of spontaneous abortion and major birth defects, although some studies have found no such link.
Depakote is approved to treat epilepsy and manic episodes of bipolar disorder. Depakote is in FDA pregnancy category D due to the potential for birth defects. In 2015, a lawsuit filed against Abbott Laboratories concerning a girl born with spina bifida after being exposed to Depakote in the womb resulted in an award of $23 million. Depakote has also reportedly been linked to cleft palate, hypoplastic right heart, and hand malformations.
Wellbutrin is approved to treat patients with major depressive disorder. The medication has been linked to an increased risk of congenital heart defects when infants are exposed to Wellbutrin during early stages of pregnancy.
Although the vast majority of births are uncomplicated, in some cases, birth injuries are due to the actions—or inactions—of medical professionals during labor. In cases where medical malpractice has resulted in infant defects or birth injuries, parents may be able to file lawsuits against the medical professionals and/or their employers whose negligence or malpractice may have resulted in the defects. Providing improper or inadequate prenatal care can cause birth defects, while failure to take appropriate action during labor can result in birth injuries.
Last updated on
FREE BIRTH INJURY AND BIRTH DEFECT LAWSUIT EVALUATION
Send your Birth Injury and Birth Defect claim to a lawyer who will review your claim at NO COST or obligation.
GET LEGAL HELP NOW
GET LEGAL HELP NOW
Birth Defects
Birth defects can be caused by a range of factors, including genetic problems or chromosome problems. But in some cases, infants are exposed to medications, chemicals or other toxins while in the womb that can affect their development. Meanwhile, injuries sustained during birth may also cause lifelong difficulties.
In cases where negligence, malpractice, or other unethical activity has occurred and that activity has resulted in birth defects, lawsuits can be filed against the people or organizations involved.
Birth Defects Linked to Medications
As of 2015, the FDA changed how drugs are labeled in relation to pregnancy. Under the new guidelines, drug labels will include three subsections—"Pregnancy," "Lactation," and "Females and Males of Reproductive Potential." Those subsections will include a summary of the risks of using a drug during pregnancy or lactation and information that will help medical professionals make prescribing decisions. This new rule replaces the letter categories, which were previously used to classify medications.
In the past, the FDA classified drugs in five pregnancy categories (this system has since been replaced but is still valid as drugs already on the market are slowly moved to the new classification):
-
Category A—Studies have not shown a risk to the fetus in the first trimester of pregnancy;
Category B—Animal reproduction studies have not shown a risk and there are no adequate studies in pregnant women;
Category C—Animal reproduction studies show an adverse effect on the fetus and there are no adequate studies in humans; benefits may outweigh the risks;
Category D—There is evidence of human fetal risk based on experience or studies in humans, but benefits may still outweigh the risks; and
Category X—Studies in animals and/or humans have shown evidence of human risk; risks outweigh potential benefits.
SSRI/SNRI Birth Defects
Some birth defects reportedly linked to SSRIs/SNRIs include persistent pulmonary hypertension of the newborn (PPHN), a disorder of the lungs in which the arteries are restricted, causing blood pressure in the pulmonary artery to drastically increase. PPHN can cause shock, heart failure, seizures, organ damage, and death.
Included among the SSRI and SNRI drugs potentially linked to birth defects are:
SSRIs SNRIs
Lyrica Birth Defects
Zofran Birth Defects
Depakote Birth Defects
Wellbutrin Birth Defects
Medical Malpractice
Birth Injury and Birth Defect Legal Help
If you or a loved one has suffered similar damages or injuries, please click the link below and your complaint will be sent to a Drug and Medical Device lawyer who may evaluate your claim at no cost or obligation.Last updated on
BIRTH INJURY AND BIRTH DEFECT LAWSUITS
- Toxic Chemical Birth Defects
- Pregabalin Linked to Increase in Birth Defects
- Zofran Birth Defects Lawsuit Information alleging heart defects and kidney malformations in newborns.
- SNRI Birth Defects allegedly cause birth defects in the children of mothers who used SNRIs in pregnancy.
- Pristiq alleging birth defects and other side effects.
- Birth Injury alleging injuries resulting at birth from medical malpractice or negligence.
- Cymbalta Alleging Cymbalta causes birth defects including as heart problems and persistant pulmonary hypertension in the newborn (PPHN) as well as severe Withdrawal Symptoms
- Lexapro Heart Birth Defect alleging Lexapro caused heart birth defects in the newborn.
- Prozac Heart Birth Defects alleging that taking Prozac during pregnancy can cause heart birth defects in the newborn.
- Zoloft Heart Birth Defects alleging that pregnant women who took the SSRI Zoloft have an increased risk of delivering a baby with heart defects.
- Effexor Heart Defects alleging that women who took Effexor while pregnant face an increased risk of having a baby with heart defects.
- Celexa Heart Birth Defects alleging Celexa, when taken during pregnancy, increases the risk a newborn will have heart defects.
- Paxil Heart Defects alleging Paxil causes heart defects in newborns.
- Symbyax Birth Defects alleging serious heart and lung birth defects Persistant Pulmonary Hypertension in Newborns (PPHN)
- SSRI Birth Defects Lawsuit allegedly causing birth defects, including heart defects and PPHN.
- Depakote alleging birth defects.
- Wellbutrin alleging that treatment with Wellbutrin may cause heart birth defects, worsening depression and/or suicidality.
BIRTH INJURY AND BIRTH DEFECT LEGAL ARTICLES AND INTERVIEWS
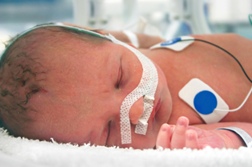
Reading Hospital Settles $32.5M Birth Injury Lawsuit

Experienced Birth Injury Attorneys Discuss Lawsuits and Settlements
Families Ask GSK to Hand Over Zofran Data

May 24, 2024
A birth injury lawsuit filed in 2022 in the Philadelphia Court of Common Pleas alleging medical malpractice has been settled for $32.5 million with Reading Hospital. The medical malpractice complaint involved several medical errors that a jury was set to hear before the settlement was reached last month. Tragically, the now 5-year-old boy suffers from permanent neurological injuries: He is nonverbal, cortically blind, has cerebral palsy and has to be tube-fed. And he will need around-the-clock care for the rest of his life. READ MORE
Experienced Birth Injury Attorneys Discuss Lawsuits and Settlements

September 23, 2023
The day your child is born should be one of the happiest days of your life. Sadly, medical complications may arise during childbirth that put your baby’s health at risk. When a doctor or a medical professional or medical facility fails to handle the situation properly, your infant may suffer injuries or illnesses resulting in permanent disability. In the worst cases, mishandled childbirth complications may result in death. Experienced birth injury lawyers at Stalwart Law know that experiencing a birth injury is nothing short of devastating, and the last thing on your mind is likely how and where to seek legal help. Even though you are left emotionally distraught and often financially hurt, attorneys Sean O’Neill and Elizabeth “Beth” Teixeira know from years of experience that sooner is better than later to help you obtain financial damages from those responsible for your child’s birth injury. READ MORE
Families Ask GSK to Hand Over Zofran Data

February 27, 2018
Washington, DC: Families of patients who allegedly suffered birth defects while taking GlaxoSmithKline’s (GSK) anti-nausea drug Zofran, and who are suing the drug maker in multi-district litigating (MDL) are asking that the drug maker hand over data stored in a drug safety database called “Argus.” READ MORE
READ MORE Drugs/Medical Settlements and Legal News
READ MORE Personal Injury Settlements and Legal News
READ MORE Personal Injury Settlements and Legal News
