LAWSUITS NEWS & LEGAL INFORMATION
Lexapro Heart Birth Defects
Lexapro (also known as escitalopram) is a type of antidepressant called a Selective Serotonin Reuptake Inhibitor (SSRI). It is used to treat anxiety and major depressive disorder. Recent studies have shown a link between SSRIs such as Lexapro and birth defects.
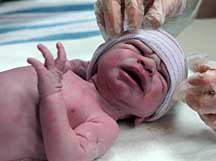 Recently, SSRIs such as Lexapro taken during pregnancy have been linked to serious birth defects in babies, including some heart problems. A Danish study found that women who used SSRIs during pregnancy were at 34 percent higher risk of delivering a baby with congenital malformations than those who did not take SSRIs during pregnancy. Furthermore, women in the study who took SSRIs similar to Lexapro during the second or third month of pregnancy had an 84 percent higher risk of having a baby with heart defects.
Recently, SSRIs such as Lexapro taken during pregnancy have been linked to serious birth defects in babies, including some heart problems. A Danish study found that women who used SSRIs during pregnancy were at 34 percent higher risk of delivering a baby with congenital malformations than those who did not take SSRIs during pregnancy. Furthermore, women in the study who took SSRIs similar to Lexapro during the second or third month of pregnancy had an 84 percent higher risk of having a baby with heart defects.
Meanwhile, a US study found that when mothers use SSRI antidepressants in the early stages of pregnancy the infant is 2.2 times more likely to have a birth defect and 2.08 times more likely to have a heart problem. The majority of heart defects involved ventricular septal defects, which can require surgery to repair.
The FDA has placed Lexapro in Pregnancy Category C, meaning that animal studies have indicated that there is a risk to the fetus, but human studies do not show that the risk of birth defects is greater than the risks involved in discontinuing medication.
So far, most SSRI studies have been done on the drug Paxil, another SSRI antidepressant. Based on those studies, the FDA has advised health care officials not to prescribe Paxil for women who are in their first three months of pregnancy or who are planning on becoming pregnant unless there are no other appropriate treatment options. More studies are required on other SSRIs that are similar to Paxil and Lexapro.
Women who are pregnant should speak with their doctor immediately about whether or not to continue taking Lexapro. In some cases, the doctor may recommend tapering off the medication in order to prevent withdrawal syndrome. However, the longer it takes to completely stop the medication, the longer the fetus is exposed to Lexapro, which could increase the likelihood of some form of heart defect in the newborn.
Class action lawsuits have been filed against the makers of SSRIs, alleging that patients were not adequately warned about the risk of birth defects when infants were exposed to the antidepressants prior to birth.
Last updated on
En Español LEXAPRO DEFECTOS DE NACIMIENTO
FREE LEXAPRO LAWSUIT EVALUATION
Send your Lexapro claim to a lawyer who will review your claim at NO COST or obligation.
GET LEGAL HELP NOW
GET LEGAL HELP NOW
Lexapro and Birth Defects
 Recently, SSRIs such as Lexapro taken during pregnancy have been linked to serious birth defects in babies, including some heart problems. A Danish study found that women who used SSRIs during pregnancy were at 34 percent higher risk of delivering a baby with congenital malformations than those who did not take SSRIs during pregnancy. Furthermore, women in the study who took SSRIs similar to Lexapro during the second or third month of pregnancy had an 84 percent higher risk of having a baby with heart defects.
Recently, SSRIs such as Lexapro taken during pregnancy have been linked to serious birth defects in babies, including some heart problems. A Danish study found that women who used SSRIs during pregnancy were at 34 percent higher risk of delivering a baby with congenital malformations than those who did not take SSRIs during pregnancy. Furthermore, women in the study who took SSRIs similar to Lexapro during the second or third month of pregnancy had an 84 percent higher risk of having a baby with heart defects.
Meanwhile, a US study found that when mothers use SSRI antidepressants in the early stages of pregnancy the infant is 2.2 times more likely to have a birth defect and 2.08 times more likely to have a heart problem. The majority of heart defects involved ventricular septal defects, which can require surgery to repair.
Lexapro Warning
So far, most SSRI studies have been done on the drug Paxil, another SSRI antidepressant. Based on those studies, the FDA has advised health care officials not to prescribe Paxil for women who are in their first three months of pregnancy or who are planning on becoming pregnant unless there are no other appropriate treatment options. More studies are required on other SSRIs that are similar to Paxil and Lexapro.
Women who are pregnant should speak with their doctor immediately about whether or not to continue taking Lexapro. In some cases, the doctor may recommend tapering off the medication in order to prevent withdrawal syndrome. However, the longer it takes to completely stop the medication, the longer the fetus is exposed to Lexapro, which could increase the likelihood of some form of heart defect in the newborn.
Lexapro Lawyer
Lexapro Heart Birth Defect Legal Help
If your baby was born with heart birth defects, and you took Lexapro while pregnant, you may need legal help. Please click the link below to send your complaint to a lawyer who will review your claim at no cost.Last updated on
LEXAPRO LEGAL ARTICLES AND INTERVIEWS
Lexapro Manufacturer Agrees to Settlement Worth Up to $10.4 Million
Women and Doctors Deal With Conflicting Information Regarding SSRI Side Effects
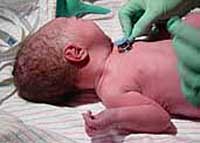
Are Americans Risking Lexapro Birth Defects Needlessly?


March 23, 2014
Plaintiffs litigating against the manufacturers of Lexapro may have lost one round of a fight accusing defendant Forest Laboratories Inc. (Forest) of misrepresentation. But another Lexapro lawsuit has been filed with regard to Lexapro and a companion drug, Celexa, amidst allegations that the manufacturer heavily promoted the drugs to be more effective than they actually are, or so it is alleged. READ MORE
Women and Doctors Deal With Conflicting Information Regarding SSRI Side Effects

September 10, 2012
When there is no consensus about the potential for Lexapro side effects, it can be difficult for patients to know if the benefits are worth the risks. After all, the risk of Lexapro birth defects has to be weighed against the benefits of taking the antidepressant. This is especially true for pregnant women, who must consider the risks to their fetus. The problem is when researchers and experts do not actually agree on the risks, leaving women and their physicians to make decisions based on incomplete information. READ MORE
Are Americans Risking Lexapro Birth Defects Needlessly?

July 30, 2012
Forest Laboratories' own exhaustive prescribing information on Lexapro (which includes a boxed warning for suicidal thoughts) admits there remains a lot of questions about Lexapro and pregnancy. "There are no adequate and well-controlled studies in pregnant women; therefore, escitalopram [Lexapro] should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus." That risk includes the potential for Lexapro birth defects that could stay with a child through his entire adult life. READ MORE
READ MORE Birth Injury and Birth Defect Settlements and Legal News
READ MORE Drugs/Medical Settlements and Legal News
READ MORE Drugs/Medical Settlements and Legal News
