 Scientific Protein (SPL) is the same company that supplied the active ingredient for the heparin distributed by Baxter International that harmed so many people across the US in the first recall.
Scientific Protein (SPL) is the same company that supplied the active ingredient for the heparin distributed by Baxter International that harmed so many people across the US in the first recall.The FDA and B. Braun Medical Inc. have issued a nationwide voluntary recall of certain lots of Heparin Sodium USP Active Pharmaceutical Ingredient (API) due to the identification of oversulfated chondroitin sulfate (OSCS) contaminant. B. Braun Medical issued the recall after being notified by SPL of a recall of one lot of API sold to B. Braun Medical.
A spokesperson for the FDA said that, "There is not a significant public health threat," but Drake disagrees. "This heparin recall may not be a widespread contamination, but it is a serious health risk to an identifiable patient population," says Drake.
After the first recall, the FDA forced the suppliers [SPL] to use Nuclear Magnetic Resonance testing to identify contaminated heparin," explains Drake. "We would hope this particular test would catch any contamination, but for whatever reason, the heparin used to make these B. Braun IV bags slipped through the cracks."
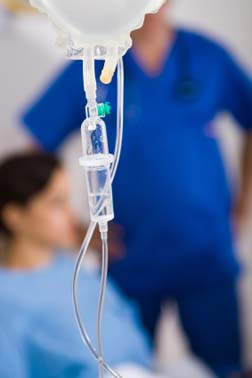
This newest heparin recall involves seven lots of Heparin IV bags. These contaminated IV drip bags were used in hospitals to treat patients who have had strokes, heart attacks or other blood vessel blockage.
Drake points out that a Heparin IV bag is given for a number of reasons, including:
- Cardiac therapy for heart attack
- Pulmonary embolism
- Deep Vein Thrombosis (DVT)
- As a safer alternative to Coumadin
Symptoms of heparin poisoning are similar to a severe allergic reaction, including a swollen throat, increased sweating, unconsciousness and even death.
Heparin Lawsuits
How big is this recall? Seven lots have been recalled, but how many bags are in a lot? The answer may have to wait until discovery. "What we do know is that these IV bags are used daily in nearly every hospital across the country," says Drake. "It is likely the contaminated IV bags were already used, so victims may have already had a reaction without knowing its cause."
READ MORE HEPARIN LEGAL NEWS
Attorney Roger Drake and Baum Hedlund are already in litigation against B. Braun, Baxter, and other heparin distributors and manufacturers for the first contamination, which occurred in 2007–08. "We have a number of contaminated heparin lawsuits already filed based on the prior recall. This new recall will create a second wave of litigation, but it is beneficial that we already have a process in place for injured patients to seek compensation."

READER COMMENTS
Harold Herrington
on
in a natural state.This cover up is appalling and i am disgusted.My Attorney claims you can hold a corporation legally responsible.I ask why not? This comes from the top,underlings do no instigate something like this.They tried a fast one in the hope of making a killing in the stock market,but instead put thousands in bodily harm with out regard.
I call that Murder in the First Degree!What Say You?
My Life Now is home bound 24/7 oxygen an medicines not yet on the Market.My Life expectancy is,What Life i am just here and no one cares.The Veterans Administration won't even acknowledge and to my knowledge never reported.To this day V.A. Cardiology Doctors claim no knowledge of recall.How Sick Is That?