LAWSUITS NEWS & LEGAL INFORMATION
Proton Pump Inhibitors Linked to Gastric Cancer, Severe Kidney Disease and Bone Fractures
Read our Proton Pump Inhibitor FAQ
By Jane Mundy
Proton pump inhibitors (PPIs) are a group of drugs mainly used to relieve symptoms of acid reflux, or gastroesophageal reflux disease. PPIs have been linked to an increased risk of bone fractures, heart attacks, cardiac birth defects (only in Prilosec), and more recently chronic kidney disease (CKD). The risk of proton pump inhibitor side effects has been found to increase with long-term use. The FDA has announced label changes that will reflect the increased risk of a proton pump inhibitor fracture, particularly PPI hip fracture. No action by the agency has yet been taken involving Proton Pump Inhibitor kidney damage or PPI renal damage.
FREE PROTON PUMP INHIBITOR LAWSUIT EVALUATION
Send your Proton Pump Inhibitor claim to a lawyer who will review your claim at NO COST or obligation.
GET LEGAL HELP NOW
GET LEGAL HELP NOW
Proton Pump Inhibitor Information
 Proton pump inhibitors are used to treat heartburn. They are also used to treat gastroesophageal reflux disease (GERD), ulcers in the stomach and small intestine, and inflammation of the esophagus.
Proton pump inhibitors are used to treat heartburn. They are also used to treat gastroesophageal reflux disease (GERD), ulcers in the stomach and small intestine, and inflammation of the esophagus.Prescription PPI medications in the proton pump inhibitor class include: (Generic name in parentheses)
- Nexium(esomeprazole)
- Dexilant (dexlansoprazole)
- Prevacid (iansoprazole)
- Prilosec (omeprazole)
- Zegerid (omeprazole)
- Protonix (pantroprazole)
- Aciphex (rabeprazole)
- Vimovo (naproxen and esomeprazole magnesium)
- Prilosec OTC (omeprazole)
- Zegerid OTC (omeprazole)
- Prevacid 24HR (iansoprazole).
Proton Pump Inhibitor Kidney Damage
Because some PPI medications are sold over-the-counter, they are potentially more at risk of being overused than prescribed PPI medications.
Chronic kidney disease is increasing in the US, with more than 20 million Americans now suffering with it, according to the American Society of Nephrology.
PPI Kidney Disease Studies
The JAMA study indicates that patients should use PPIs only when medically necessary. Dr. Morgan E. Grams, a kidney specialist at Johns Hopkins and another author of the JAMA study told CBS News that patients who require a long course of PPIs should routinely monitor their kidney function.
2015: Two studies suggested a link between PPIs and CKD. Dr. Pradeep Arora, a nephrologist and associate professor at the SUNY Buffalo School of Medicine and Biomedical Science in Buffalo, NY., said "It is very reasonable to assume that PPIs themselves can cause chronic kidney disease." Researchers also found that people taking a PPI had nearly twice the risk of dying prematurely.
The second study was conducted by researchers at Royal Brisbane and Women's Hospital in Australia, and Johns Hopkins University in Baltimore. They followed more than 10,000 adults with normal kidney function from 1996 to 2011 and found PPI users were up to 50 percent more likely to develop chronic kidney disease than non-PPI users.
2009: A study conducted in Copenhagen and published in Gastroenterology reported that PPI use may produce a "rebound"acid effect, likening the condition to "dependency"on the medications. Researchers found that patients have almost impossible difficulties in stopping PPIs because the medications eventually cause the same problems they are supposed to treat. This phenomenon, called "rebound acid hypersecretion," can occur after taking prescription-strength PPI medication for just two months. Researchers concluded that this rebound effect can continue for up to three months after discontinuing the PPI.
PPI Kidney Disease Lawsuits
In 2017, a lawsuit files against AstraZeneca and Proctor & Gamble, alleging that use of Nexium and Prilosec caused a woman to develop chronic kidney disease. The lawsuit, filed by Wanda Stafford, claims the companies failed to warn consumers about the risk of side effects linked to the medications. The lawsuit seeks punitive and compensatory damages.
Proton Pump Inhibitor Bone Fractures
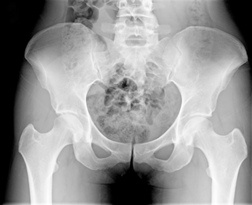 Long-term and/or high dose use of proton pump inhibitors has been linked to an increased risk of fractures, including hip, wrist and spine fractures. A study examining 145,000 people in England found patients who used the medications for more than a year had a 44 percent higher risk of hip fracture than people who did not use the medications. The largest risk was seen in people who took high doses of the drugs for more than a year. In that group, the risk of hip fractures was twice as great as patients who did not use the medications.
Long-term and/or high dose use of proton pump inhibitors has been linked to an increased risk of fractures, including hip, wrist and spine fractures. A study examining 145,000 people in England found patients who used the medications for more than a year had a 44 percent higher risk of hip fracture than people who did not use the medications. The largest risk was seen in people who took high doses of the drugs for more than a year. In that group, the risk of hip fractures was twice as great as patients who did not use the medications.An editorial accompanying studies in the May issue of the Archives of Internal Medicine noted that proton pump inhibitors have US sales of $13.9 billion annually. The same editorial argues that proton pump inhibitors have been over-prescribed for inappropriate uses.
Proton Pump Inhibitor Warning
Over-the-counter proton pump inhibitors are recommended only for 14 days of continuous use.
Approximately 15 million people in the US alone are taking either over-the-counter or prescription PPI medications, according to CBS News. In 2013, the PPI Nexium was the second-best selling drug in the pharmaceutical industry, generating over $6.1 billion in sales worldwide for AstraZeneca, as reported by Medscape.
Proton Pump Inhibitor (PPI) Legal Help
If you or a loved one has suffered kidney disease or bone fractures as a result of using any of the above mentioned products, please click the link below and your complaint will be sent to a Drug and Medical Device lawyer who may evaluate your claim at no cost or obligation.
Last updated on
PROTON PUMP INHIBITOR LAWSUITS
- Proton Pump Inhibitors Linked with Higher Rates of Gastric Cancer
- Prevacid Side Effects could include an increased risk acute kidney injury and chronic kidney disease
- Prilosec PPI Side Effects including severe kidney disease, bone fractures, heart attacks and cardiac birth defects
- Nexium Use and Increased Risk for Fracture
- Plavix and Nexium A Potentially Dangerous Combination
- AstraZeneca, Nexium Side Effects alleging severe kidney disease and risk of bone fractures
PROTON PUMP INHIBITOR LEGAL ARTICLES AND INTERVIEWS
Recent Studies Show Other Side Effects, including certain Cancers, with Long-term PPI Use

PPI Wrongful Death Lawsuit
Proton Pump Inhibitor Maker Facing Wrongful Death Lawsuit


June 3, 2021
For more than a decade, studies have shown that proton pump inhibitors (PPIs) are associated with serious side effects, including kidney failure and other injuries. Researchers now add to “other” injuries biliary track cancer, gallbladder cancer and cholecystitis (inflammation of the gallbladder) and might be carcinogenic. READ MORE
PPI Wrongful Death Lawsuit

April 16, 2021
A wrongful death lawsuit has been filed against AstraZeneca, Merck & Co., Procter & Gamble and other manufacturers of proton pump inhibitor (PPI) drugs. Plaintiff Daisy Perez claims her husband, a New Mexico man, died of metastatic gastric cancer after taking Nexium and Prilosec. READ MORE
Proton Pump Inhibitor Maker Facing Wrongful Death Lawsuit

February 18, 2021
Proton Pump Inhibitor (PPI) manufacturers and distributors AstraZeneca and Merck are facing a wrongful death lawsuit alleging the side effects of Nexium caused stomach cancer, which lead to death. READ MORE
MORE PROTON PUMP INHIBITOR LEGAL NEWS
- Proton Pump Inhibitors – More Harm than Good, especially in COVID-19 Patients
- “Acid Reflux Drugs Caused my Type 2 Diabetes”
- Regular Acid-reflux Drug Use Linked to Higher Risk of type 2 Diabetes
- Study Further Links Proton Pump Inhibitors to Gastric Cancer and Conflicts with FDA study, Lawsuits to Follow
READ MORE Drugs/Medical Settlements and Legal News
READ MORE Personal Injury Settlements and Legal News
READ MORE Personal Injury Settlements and Legal News

READER COMMENTS
Michael Blackburn
on
Sharon
on
Pete
on
WB Pennsylvania
on
Diagnosed with Pernicious Anemia 2011; Diagnosed with Iron Deficiency Anemia 07/23/2014; Diagnosed 09/18/2013 with B12 Deficiency Anemia; On 12/11/2014 diagnosed with GERD; On 02/05/2016 diagnosed with Gastroesophageal Reflux and on 12/05/2016 diagnosed with Eosinophilic Esophagitis. After referred to Allergist/Immunologist and tested for food allergies shown no allergies except of penicillin since birth. Complete six Elimination Diet: No Dairy, eggs, soy, tofu, wheat, peanuts, tree nuts. A OVO-Vegetarian (Lactose intolerant since 2005 due to surgical removal of entire large intestine and I only eat egg whites.) Since age 16, I have been a Pescatarian aka Pescetarian vegetarian. No allergies towards six elimination diet foods eliminated for twelve weeks. Prescribed by Gastrologist following medications: Budesonide Inhalation Suspension 0.5 mg/2ml twice daily; Flovent HFA 110 mcg Inhaler not to use like an Asthma patient Inhale puff by mouth 2 times daily beginning 02/05/2016; Fluticasona Prop 50 mg Spray twice in each nostril 2x's daily (11/10/2015); Omeprazole 20 mg 2 capsules 2x's daily. Allergist agreed with gastrologist to use all of the above. In September, 2016 started getting severe abdominal pain, weakness/fatigue, loss of appetite, nausea/vomiting, metal/metallic taste in mouth, more trouble sleeping and concentrating/confusion, swelling ankles, numbness in toes especially right foot, pain lower back, not urinating normal. With GERD developed severe hoarseness, coughing, sore throat, chest pain, acid reflux, regurgitating, followed from dry heaving because I wasn't eating due to acid reflux, sour taste, metal metallic taste. Sick to stomach. Had Abdominal Ultrasound (Normal) 09/2016 for abdominal pain. Saw female Gyno did vaginal ultrasound and pap test came back normal. Continue to experience abdominal pain. On 03/31/2017: Saw primary care doctor's PA-C ran blood work: ALT and AST high levels did HEP Blood work but came back Negative. Creatinine levels, RDW, MPV, Platelet Volume were all High; High Cholesterol (Put me on Lipitor) but I don't eat prepackaged foods and no junk food whatsoever not even a candy bar because I have chronic migraines of 15 or more a month (No wine, cheese, chocolate, or MSG). My , Triglycerides and RDW/MPV were High and Anion Gap was High. HDL, Chloride, Creatinine all Low Levels. LDL Calc was uninterpretable. TSH was High and T4 was Low. E Glom Filt Rate was Low. My GFR was 88-128. And my BUN level was extremely high. Primary Care PA-C said lay off Tynenol (I cannot take OTC Pain medications must be prescribed only Tramadol or a Morphine Drip). For Migraines I administer myself Imitrex Injections when severe. On 04/12/2017 had Renal/Kidney/Liver Sonograms. Fasted for 12 hours no food, no beverages and my kidney bladder showed full. I don't urinate as much. Last bloodwork on 04/07/2017 showed Creatinine High 1.4 mg/dl. Chloride 97 mol/l Low. Anion Gap 22 mmol/l High; EGR 45.8 to 43. 5 Low. Hemoglobin and Glucose showed no diabetes and low blood pressure. Findings: Kidney disease push fluids doctor said and take Lipitor once a day. On 05/04/2017 messaged Primary PA-C and told her I have been in bed 24/7 only leaving for bloodwork and ultrasounds/sonograms. No appetite, nausea, vomiting, weakness/fatigue/too weak to stand up and walk downstairs even too weak to shower, facial puffiness, more than troublesome sleep issues even more severe than being having chronic insomnia, not urinating much, trouble concentrating/confusion, chest pain, acid reflux worsened since stop taking omeprazole, now taking OTC Tums. When my daughter visits for weekends: She forces me to eat something daily and drink lots of fluids but I vomit. No appetite. I just lay in bed 24/7 lost job due to kidney disease, chronic migraines (no sleep, not eating, causes migraines); chronic sinusitis. Bloodwork shows Stage 3 Kidney Disease: I have all the signs and symptoms but all my primary care PA-C is doing is bloodwork for both Kidney's and Liver as well as Abdominal Ultrasounds and then Renal and Abdominal Sonograms. Instructions stop taking Omeprazole (Which I did over a month or two ago), stop pain medications (Which I don't have unless prescribed check with pharmacy), no OTC pain medications like Tynenol can't take OTC NSAIDs make me sick to stomach, no alcohol (Liquor store products makes me sick to my stomach: Don't drink hard liquor or wine). Been drinking cases of Gatorade to push fluids. I hate water. I have been complaining of pain since September, 2016 and learned about unknown side effects and supposed medically community were not aware of developing CKD, Kidney Failure, Heart Failure, etc. Now I recently learned through my own investigation which is currently under legal review is Lipitor used to treat high cholesterol. My doctor or pharmacist mentioned side effects could lead also to Kidney Disease, Diabetes, Heart Issues. I have an underactive thyroid since age 16. Don't know where to go from here? I have to work to pay my bills. Been off work since January, 2017 no money for food, medications (I must take my thyroid med every day and have a supply of Migraine injections for emergencies and cannot afford) no money for food for myself and my dependent daughter. No job we will be homeless, lose my car, etc. Am I eligible to be represented to sue the manufacturer for all of the above? Can your law firm represent me as well as other victims?
Lillian Tucker
on
Prilosec and then on omeprazole because his insurance wouldn't
He also broke his hip and had surgery on it.
Wayne
on
Bill Stone
on
Donald Sass
on
Michael Powell
on
Ashley Jump
on
David V.
on
Nexium(esomeprazole)
Dexilant (dexlansoprazole)
Prevacid (iansoprazole)
Prilosec (omeprazole)
Zegerid (omeprazole)
Protonix (pantroprazole)
Aciphex (rabeprazole)
Vimovo (naproxen and esomeprazole magnesium)
Prilosec OTC (omeprazole)
Zegerid OTC (omeprazole)
Prevacid 24HR (iansoprazole).
Jennie Drummond
on
I never recovered from any of this!! I have lost about 40 lbs. over a period of time and all muscle mass. I have pernicious anemia and have terrible bones according to results of density test ! If there is help, I could surely use it!! I have lost all .
elishaargo
on
Florida
on