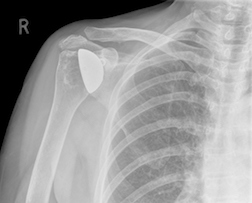
 Every surgery has a risk of failure, and because shoulder joint replacements and reverse total shoulders are commonly performed, a substantial number of patients have experienced shoulder replacement arthroplasty failures for numerous reasons. The UW Orthopaedics & Sports Medicine (2013) states that “often these failures can be improved by rehabilitation or revision surgery.” A more recent study indicates that certain patients should opt for rehabilitation rather than revision.
Every surgery has a risk of failure, and because shoulder joint replacements and reverse total shoulders are commonly performed, a substantial number of patients have experienced shoulder replacement arthroplasty failures for numerous reasons. The UW Orthopaedics & Sports Medicine (2013) states that “often these failures can be improved by rehabilitation or revision surgery.” A more recent study indicates that certain patients should opt for rehabilitation rather than revision.A study in the Journal of Bone & Joint Surgery (Jun 7, 2017) reports that patients with higher body mass index (BMI) are more likely to require revision surgical procedures and have postoperative complications after shoulder arthroplasty. And complication risk increases with increasing BMI. "These findings support the notion that increasing BMI increases the stress on the implant, leading to higher rates of mechanical implant failure," Dr. Wagner and coauthors at the Mayo Clinic report. The researchers suggest that the increased infection risk is likely related to immune system changes and to "dead space" created by excess fatty tissue.
The Mayo Clinic researchers studied data on 4,567 shoulder arthroplasties performed between 1970 and 2013. Forty-three percent of the patients were obese (with a BMI of 30 or higher). The researchers analyzed the relationship between BMI and different types of complications and found that 302 patients required revision surgery due to mechanical failure, loosening of the implant, or other causes.
The most common complications after reverse shoulder arthroplasty included instability, periprosthetic fracture, infection, component loosening, neural injury, acromial and/or scapular spine fracture, hematoma, deltoid injury, rotator cuff tear, and venous thromboembolism (VTE). The study pointed out that diagnosing infection after shoulder arthroplasty “remains a challenge”.
Although advancements in procedures and new implants have made shoulder replacement surgery much less invasive, with less pain and shorter recovery time, infection and device failure is still a risk. A number of shoulder replacement devices have been recalled, including the Zimmer Biomet Comprehensive Reverse Shoulder, which was approved by the FDA without any human clinical trials (it was approved by way of the 510K process) and pulled from the market in 2010 after complaints of fracturing. Fractures that result in revision surgeries could cause “serious adverse health consequences such as permanent loss of shoulder function, infection, or rarely, death,” according to the FDA. They classified the recall as a “Class I,” the most serious type of medical device recall.
READ MORE BIOMET SHOULDER REPLACEMENT LEGAL NEWS
According to the Zimmer website, less than 1.9% of patients get an infection in the first two years. This statistic was taken from the Journal of Bone Joint Surgery back in 2002. Needless to say, it looks like Zimmer hasn’t updated its site with recent studies.

READER COMMENTS
TRINA JACQUILLARD
on
Annie M Wagner
on
I didn't have a shoulder replacement but I did haveArthroscopy surgery done on my right shoulder,after having so many injection.Which I am still bother by it today.The surgery was done 2009