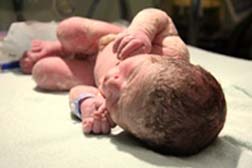
 A study published in Neurology (online May 2016) concluded that “there may be an increased risk for major birth defects after taking pregabalin during the first trimester of pregnancy” but the authors also wrote they can’t draw “definitive conclusions” from the study “since many of the women were taking other drugs that could have played a role in the birth defects and because the study was small.” According to FiercePharma, Pfizer agreed the study was small, and noted that it didn’t take into considerations other medical conditions. In an email to FiercePharma, the drug company said that “The women taking Lyrica had higher rates of smoking and diabetes, all of which can negatively affect pregnancy outcomes.”
A study published in Neurology (online May 2016) concluded that “there may be an increased risk for major birth defects after taking pregabalin during the first trimester of pregnancy” but the authors also wrote they can’t draw “definitive conclusions” from the study “since many of the women were taking other drugs that could have played a role in the birth defects and because the study was small.” According to FiercePharma, Pfizer agreed the study was small, and noted that it didn’t take into considerations other medical conditions. In an email to FiercePharma, the drug company said that “The women taking Lyrica had higher rates of smoking and diabetes, all of which can negatively affect pregnancy outcomes.”Of course studies are not conducted on pregnant women. The researchers said that most of the women in the study started taking Lyrica before they became pregnant but stopped taking it about six weeks into their pregnancy. In Pfizer’s defense, 22 of these women were also taking another anti-seizure drug.
But the study showed that birth defects linked to Lyrica included heart defects and problems with the central nervous system or other organs. Women taking Lyrica were six times more likely to have a baby with a major defect in the central nervous system than women not taking the drug, according to the researchers.
So far, the FDA has not asked Pfizer to change its label and warn pregnant women about possible birth defects. But it warns men to “Talk to your healthcare provider if you plan to father a child” and that “Animal studies have shown that pregabalin, the active ingredient in Lyrica, made male animals less fertile and caused sperm to change. Also, in animal studies, birth defects were seen in the offspring (babies) of male animals treated with pregabalin. It is not known if these problems can happen in people who take Lyrica.”
Animal studies on the effects of fetal exposure to drugs often support claims in birth defect lawsuits. For instance, Results of recent Japanese studies showed that pregnant animals who were exposed to Zofran subsequently gave birth to offspring with heart defects.
READ MORE LYRICA LEGAL NEWS
Hundreds of people in the US have filed Zofran birth defect lawsuits claiming that exposure to the anti-nausea drug caused birth defects in their children. Will Lyrica follow suit? Attorneys are currently investigating Lyrica claims. Stay tuned…
