 Katy (not her real name) says one of her daughters was prescribed Accutane to treat acne just before it was recalled in 2009 and her other daughter was also prescribed Accutane, but her pharmacist dispensed Claravis, a generic Accutane medication. (There are a number of generic versions of Accutane (isotretinoin) on the market, including Amnesteem and Sotret, and all of them are rated "AB" by the FDA, meaning that they should be equivalent to the brand name, i.e., Accutane.)
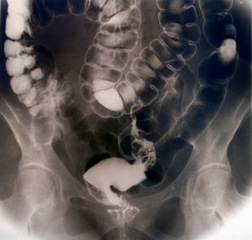
Katy (not her real name) says one of her daughters was prescribed Accutane to treat acne just before it was recalled in 2009 and her other daughter was also prescribed Accutane, but her pharmacist dispensed Claravis, a generic Accutane medication. (There are a number of generic versions of Accutane (isotretinoin) on the market, including Amnesteem and Sotret, and all of them are rated "AB" by the FDA, meaning that they should be equivalent to the brand name, i.e., Accutane.)"My youngest daughter developed bowel problems last fall and when the pain and diarrhea didn't abate I took her to a gastroenterologist," says Katy. "Her blood tests show markers for Crohn's disease and that has really got me worried. She is in university and it is already affecting her daily activities and sleep patterns—Crohn's disease is going to change her life, to her detriment.
"It wasn't until now that I connected the dots. I was trying to figure out when she first developed intestinal problems and then I realized she finished the course of Claravis at the same time—Bingo! I immediately called my daughter but she had already put two and two together.
"As for my other daughter who took Accutane, she also suffers from bowel problems but she handles it differently—even though her symptoms are severe, she isn't so emotionally affected. Still, I am having her checked for Crohn's as soon as possible, but the Statute of Limitations has run out on her.
"Another reason it took me so long to link Accutane to IBD and Crohn's is because I mainly heard about the emphasis on Accutane birth defects. And the government's iPLEDGE program completely shut my mind to the fact that Accutane had side effects other than birth defects.
READ MORE ACCUTANE IBD LEGAL NEWS
Katy understands that the Supreme Court decision regarding a generic drug has kept attorneys from proceeding on these kinds of claims, but a recent New Jersey Supreme Court case gives her hope regarding an Accutane lawsuit. The Court found for the plaintiff, determining that Roche, the Accutane manufacturer, had insufficient warnings on its patient literature and the plaintiff's claim was not subject to the Statute of Limitations.

READER COMMENTS
Chunghoe
on
Jason Binnes
on