Protopic Cream and Skin Cancer
Protopic (tacrolimus), is a topical ointment used in the treatment of eczema. Protopic was approved by the FDA in December 2004. Protopic should be used only on areas of skin that have eczema, and should not be allowed to contact normal skin.
In March, 2005 the FDA issued an alert warning doctors and patients about the increased risk of cancer from using Protopic. Nineteen cases of Protopic-related cancer had been reported as of December 2004: three children and 16 adults, two of whom died. Some of the victims were diagnosed with cancer within as little as 21 days after starting treatment with Protopic, but the average time to diagnosis was five months.  Animal studies on rats, mice and monkeys revealed a link between Protopic use and skin cancer, and some patients using the cream have developed the disease. Animal studies also showed that the higher the dose of Protopic used, the higher the rates of cancer became.
Animal studies on rats, mice and monkeys revealed a link between Protopic use and skin cancer, and some patients using the cream have developed the disease. Animal studies also showed that the higher the dose of Protopic used, the higher the rates of cancer became.
It is believed that Protopic causes skin cancers and lymphoma by suppressing the body's immune system. Protopic is also associated with leukemia, Hodgkins and non-Hodgkins lymphoma cancers in children, and breast cancer.
Evidence began to appear that people using Protopic were developing non-Hodgkins lymphoma, squamous cell disease and malignant lymphomas. As early as the year 2000, lymphoma was identified as a serious side effect of Protopic use. An FDA Medical Examiner recognized with Protopic:
The high and progressively increasingly hazard rates of lymphodenopathy in this analysis raised some concerns, especially when considered together with the preclinical toxicology studies showing high incidence of lymphoma in mice treated topically with 0.1% Tacrolimus ointment.Lymphodenopathy is usually an indication of some underlying disease, and can lead to malignancy. The manufacturers of Protopic have from the outset been marketing the drug without adequate cancer warnings despite their knowledge of the lymphoma risks. Their PDR references and product inserts for physicians did not contain this type of cancer risk information.
Physicians have now been warned to give their patients Protopic only for short-term applications, and to only use the drug if other eczema treatments have failed. Protopic should not be used in children under the age of two, and anyone with a compromised immune system should not use Protopic at all.
Protopic Cancer Articles
Protopic and Elidel Prescribed to Kids Despite Cancer RiskWhen a significant side effect is linked to a drug, the Food and Drug Administration warns that the benefits of taking a drug should be weighed against the risks; however, in the cases of Elidel and Protopic, which have been linked to cancer, there is almost no way that the possible benefits could ever be greater than the risks.
Cancer Connection: Protopic and Elidel
My wife broke out in nasty rashes and the doctor assumed it was eczema so she was prescribed Elidel," says Fred Laincz. "Her rashes cleared up but at the same time she became very sick." Betsy Laincz, 59, was diagnosed with liver cancer and passed away last month.
Protopic Warning Linked to Cancer
Cancer Risks Associated with Protopic and Elidel Long Suspected
Protopic linked to Child's Leukemia
Elidel and Protopic: File Sooner than Later
Protopic Warning Too Late
Protopic ointment linked to Lymphoma Cancer
Protopic In the News
MAY-14-07: A woman who used Protopic to treat a skin condition on her breast developed breast cancer and required a mastectomy. She says she was never warned that the medication could cause cancer.APR-27-07: Protopic, a drug used to treat skin conditions, has been linked to cases of Hodgkin's lymphoma. In one case, a young boy used Protopic for a year before being diagnosed with stage three cancer. In 2003, an FDA advisory panel decided that the public should be made aware of the risk of cancer associated with Protopic, but a warning was not made public for over two years.
MAR-06-07: Despite the black box warnings ordered by the FDA last year, drug companies maintain that, used as directed, Protopic is safe and there is no conclusive evidence linking the drug to cancer.
FEB-21-07: Several wrongful death and injury lawsuits involving Protopic have recently been filed as victims fall prey to cancer. [
DEC-04-06: Deadline for legal action against the manufacturers of Protopic is approaching. [EWORLDWIRE: PROTOPIC DEADLINE]
NOV-29-06: Lawyers investigate the link between Protopic and cancer.
NOV-28-06: Consumer Reports says some people may develop skin cancer or lymphoma after using Protopic ointment. [HEARTLAND NEWS: PROTOPIC CANCER]
OCT-20-06: Protopic has "black box" warning telling users about the possibilities of getting skin cancer from using the drug. [
APR-27-06: Health Canada warns people using Protopic ointment that there is a potential cancer risk. [
MAR-10-05: FDA's Pediatric Advisory Committee reviewed tests that linked Protopic to an increased risk of skin cancer and non-Hodgkin's lymphoma. [FOX NEWS; PROTOPIC SKIN CANCER]
Protopic Skin Cancer
If you have developed skin cancer, breast cancer or other cancer and have been using Protopic, a lawyer may be able to help you. Please click the link below to send your story to an attorney who will evaluate your claim at no charge.Last updated on
LEGAL ARTICLES AND INTERVIEWS
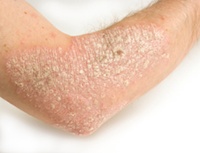
Protopic and Elidel Prescribed to Kids Despite Cancer Risk

READER COMMENTS
Constance Sullivan
on
Michelle Valentine
on