Zimmer Durom Cup HIP REPLACEMENT
The Zimmer Durom Cup is a newer type of hip replacement component, made with a single piece of material. Since it was introduced to the US market in 2006, the cup has been used for total hip replacement (known as hip arthroplasty) in more than 12,000 patients, but many individuals have reported severe pain and the need for additional hip surgeries after having the Zimmer Durom Cup hip replacement implanted. According to the manufacturer, doctors reported post-hip surgery problems "requiring revision" 5.7 percent of the time. In other words, at least 684 patients may require additional hip surgeries after having a Zimmer hip replacement. Some patients have also experienced problems with the Zimmer NexGen CR-Flex knee replacement component.
FREE ZIMMER DUROM CUP LAWSUIT EVALUATION
GET LEGAL HELP NOW
Zimmer Durom Cup Hip Replacement
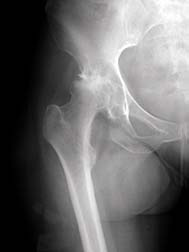 In July 2008, after reviewing data on more than 3,000 cases, Zimmer, the world's largest manufacturer of hip replacement parts, suspended sales of the Durom Acetabular Component (Durom Cup) until it can update the label with instructions about special surgical techniques to be followed during total hip replacements.
In July 2008, after reviewing data on more than 3,000 cases, Zimmer, the world's largest manufacturer of hip replacement parts, suspended sales of the Durom Acetabular Component (Durom Cup) until it can update the label with instructions about special surgical techniques to be followed during total hip replacements.
Because the Indiana-based manufacturer says it did not uncover evidence of any manufacturing defect or design defect, a recall was not issued.
Zimmer Durom Cup Lawsuits
Class action and individual lawsuits have been filed against Zimmer Holdings Inc., claiming it failed to provide proper warnings, instructions and training to surgeons.
In April 2008, Dr. Larry Dorr, a prominent orthopedic surgeon, wrote an open letter to members of the American Association of Hip and Knee Surgeons complaining of Zimmer Durom Cup problems: of 165 Zimmer Cup implants he performed, 14 were "revised or required revision" within the first two years after surgery. Dr. Dorr indicated that the Cup was defective: the fixation surface on the cup was problematic and a circular cutting surface on the periphery of the cup prevented it from fully seating.
One month later, Zimmer sent a letter to healthcare providers indicating they were initiating an investigation into the complaints of Zimmer Durom Cup complications, which led to the suspension of sales in July.
According to a 07/30/10 report in The New York Times Zimmer blamed the problem on Dr. Door's technique and provided the FDA with information indicating that the hip replacement worked as intended. The FDA investigation was then closed.
Senator Seeks Zimmer Information
In July 2010, Senator Charles E. Grassley (R, Iowa) asked Zimmer Holdings to provide information about how it handles complaints about product problems from medical consultants and how it tracks long-term performance of its devices. The request was made after a 06/20/10 article in The New York Times, which outlined two disputes between Zimmer consultants and the device maker—both of which involved consultants claiming there were problems with Zimmer products. In both cases, Zimmer reportedly blamed the technique used to implant the device for the failure.
Zimmer responded to Grassley's request saying it "welcomed the opportunity" to speak with Grassley.
Zimmer advises patients who have received the Durom Cup to contact their doctor if they experience pain more than three months after surgery.
Zimmer Durom Cup Legal Help
If you or a loved one has suffered damages as a result of a Zimmer Durom Cup, you may qualify for damages or remedies that may be awarded in a possible class action or lawsuit. Please click the link below to submit your complaint to a lawyer for a free evaluation.Last updated on
ZIMMER DUROM CUP LEGAL ARTICLES AND INTERVIEWS
Widow Launches Zimmer Hip Lawsuit
Zimmer Inc. Earns Profit despite Costs of Zimmer Hip Lawsuits
READ MORE Defective Products Settlements and Legal News
READ MORE Drugs/Medical Settlements and Legal News

READER COMMENTS
Cheryl Robb
on
Thank you so very much, Dr Dorr! Sorry to hear you are still able to ruin people’s lives......
Jozetta Bullock
on
Greenville, WI
on
2nd hip replaced 1st hip May 2015. Before surgery tested positive with MRSA in nose. After surgery felt good and walking good. End of June 2015 got really sick with fever, chills etc. Put in hospital and removed hip and scrapped out infection. Put in a cement block in hip with antibiotic. Now I live like this for aprox 2 months or more until infection is gone. Then I go for the 3rd hip all three on my write side..
Massachusetts
on
Anonymous
on
I see so much about other brands and part as in the discontinued cup, but I can't imagine that I am the only one with these issues.
Indiana
on
Washington
on
Mississippi
on
Arizona
on
California
on
Alabama
on
Maryland
on
Texas
on
Have been unable to walk since 2001, Had a heart attack trying to ambulate. Have had therapy for approximately 6 years Cannot express the depression I fell into.
My husband had to take an early retirement due to the fact I could not be left alone. It has ruined both of our lives. Also having to replace my right knee again. There is not an amount I can ask for that will cover the damage I have endured.