LAWSUITS NEWS & LEGAL INFORMATION
Wellbutrin Side Effects
Were you looking for Paxil Heart Defects lawsuits?
Wellbutrin (known generically as bupropion hydrochloride) is approved to treat patients with major depressive disorder. Wellbutrin side effects may include serious health events, such as Wellbutrin birth defects in infants when taking Wellbutrin while pregnant. Those birth defects include potentially serious congenital heart defects.
An article published in the July 2010 issue of American Journal of Obstetrics & Gynecology suggests an increased risk of congenital heart defects when infants are exposed to Wellbutrin during early pregnancy. Specifically, an association was found between the use of Wellbutrin during the first trimester and left outflow tract defect, which can restrict blood flow to the infant's body from the left chambers of the heart. Infants born with left outflow tract defect often require surgery to correct the problem.
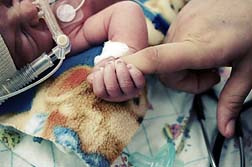 Left outflow heart defects include coarction of the aorta, hypoplastic left heart syndrome, pulmonary stenosis, aortic stenosis, teralogy of fallot ventricular septal defects and atrial septal defects.
Left outflow heart defects include coarction of the aorta, hypoplastic left heart syndrome, pulmonary stenosis, aortic stenosis, teralogy of fallot ventricular septal defects and atrial septal defects.
The study involved data from 12,700 children born in the US between 1997 and 2004. Researchers found that infants exposed to bupropion during the first trimester were more than two times as likely to have a congenital heart problem. At the time, researchers recommended further studies be conducted.
Wellbutrin is classed as Pregnancy Category C, meaning there are studies conducted in animals that demonstrate a potential risk to the fetus, but there are currently not enough human studies.
In 1996, the FDA approved a sustained-release tablet of bupropion hydrochloride called Wellbutrin SR and, in 2003, approved an extended-release tablet known as Wellbutrin XL—all manufactured by GlaxoSmithKline. The drug was pulled a year later due to the high number of seizures associated with Wellbutrin side effects, but in 1989 federal reviewers allowed it back on the market after determining that reports of seizures were dose specific--Glaxo lowered the daily dosage of the drug.
Wellbutrin belongs to the aminoketone class. It is structurally and chemically unlike other antidepressants, such as Prozac, Paxil and Zoloft. The mechanism of its therapeutic actions is not well understood, but it does appear to block dopamine uptake. Wellbutrin is sometimes used "off-label" for ADHD, weight loss, and even sexual dysfunction.
 In 2004 GlaxoSmithKline, along with the FDA, issued a Wellbutrin warning to healthcare professionals stating in part, that patients being treated with antidepressants may "experience worsening of their depression and/or the emergence of suicidal ideation and behavior". Professionals were encouraged to watch closely for signs of "clinical worsening and suicidality" in their patients, particularly when starting or stopping the drug, as well as when dosages are changed.
In 2004 GlaxoSmithKline, along with the FDA, issued a Wellbutrin warning to healthcare professionals stating in part, that patients being treated with antidepressants may "experience worsening of their depression and/or the emergence of suicidal ideation and behavior". Professionals were encouraged to watch closely for signs of "clinical worsening and suicidality" in their patients, particularly when starting or stopping the drug, as well as when dosages are changed.
In October 2004 the FDA approved the following for inclusion on a "black box" warning: Antidepressants increase the risk of suicidal thinking and behavior (suicidality) in children and adolescents with MDD and other psychiatric disorders." Wellbutrin is not approved by the FDA to treat depression in children. Patients, especially those younger than 24 years of age, can experience worsening suicidal thoughts and behaviors.
Like Wellbutrin, Zyban is known generically as bupropion hydrochloride. Zyban is used as a smoking cessation drug and Zyban side effects may be similar to Wellbutrin side effects. On July 1, 2009, the FDA required the maker of Zyban to include a boxed warning about the risk of mental health events. Such Zyban adverse effects could include changes in behavior, depressed mood, hostility and suicidal thoughts, in patients when taking Zyban.
Last updated on
FREE WELLBUTRIN LAWSUIT EVALUATION
Send your Wellbutrin claim to a lawyer who will review your claim at NO COST or obligation.
GET LEGAL HELP NOW
GET LEGAL HELP NOW
Wellbutrin Birth Defects
 Left outflow heart defects include coarction of the aorta, hypoplastic left heart syndrome, pulmonary stenosis, aortic stenosis, teralogy of fallot ventricular septal defects and atrial septal defects.
Left outflow heart defects include coarction of the aorta, hypoplastic left heart syndrome, pulmonary stenosis, aortic stenosis, teralogy of fallot ventricular septal defects and atrial septal defects.The study involved data from 12,700 children born in the US between 1997 and 2004. Researchers found that infants exposed to bupropion during the first trimester were more than two times as likely to have a congenital heart problem. At the time, researchers recommended further studies be conducted.
Wellbutrin is classed as Pregnancy Category C, meaning there are studies conducted in animals that demonstrate a potential risk to the fetus, but there are currently not enough human studies.
Wellbutrin Side Effects
Wellbutrin belongs to the aminoketone class. It is structurally and chemically unlike other antidepressants, such as Prozac, Paxil and Zoloft. The mechanism of its therapeutic actions is not well understood, but it does appear to block dopamine uptake. Wellbutrin is sometimes used "off-label" for ADHD, weight loss, and even sexual dysfunction.
Wellbutrin FDA Warning
 In 2004 GlaxoSmithKline, along with the FDA, issued a Wellbutrin warning to healthcare professionals stating in part, that patients being treated with antidepressants may "experience worsening of their depression and/or the emergence of suicidal ideation and behavior". Professionals were encouraged to watch closely for signs of "clinical worsening and suicidality" in their patients, particularly when starting or stopping the drug, as well as when dosages are changed.
In 2004 GlaxoSmithKline, along with the FDA, issued a Wellbutrin warning to healthcare professionals stating in part, that patients being treated with antidepressants may "experience worsening of their depression and/or the emergence of suicidal ideation and behavior". Professionals were encouraged to watch closely for signs of "clinical worsening and suicidality" in their patients, particularly when starting or stopping the drug, as well as when dosages are changed.Wellbutrin Depression and Suicidal Thoughts
Zyban Side Effects
Wellbutrin Legal Help
If you or a loved one has experienced side effects from the use of Wellbutrin or if you have taken Wellbutrin while pregnant and your child has suffered from a birth defect, please click the link below to submit your complaint and we will have a lawyer review your case.Last updated on
WELLBUTRIN LEGAL ARTICLES AND INTERVIEWS
Study Suggests Link between Wellbutrin and Birth Defects

How Much 'Well' is Associated With Wellbutrin?

Former Glaxo Executive Indicted

August 6, 2011
It has been just over a year since a study was published in the American Journal of Obstetrics & Gynecology suggesting a link between the use of Wellbutrin early in pregnancy and the development of Wellbutrin birth defects. The study concluded that more data would be needed to confirm potential Wellbutrin side effects on infants. Additional studies have not been conducted, but the FDA has Wellbutrin in pregnancy category C and suggests that it should be used during pregnancy only if the potential benefit justifies the potential risk. READ MORE
How Much 'Well' is Associated With Wellbutrin?

July 16, 2011
There appears to be some disconnect as to just what FDA pregnancy category Wellbutrin is assigned to. Most sources hold that the drug is categorized as 'C', which is described by the Journal of Counseling and Development (JCD) as posing a potential risk to the fetus that cannot be ruled out, due to the fact that animal-based studies have shown adverse effects on the fetus, but that no well-controlled studies on pregnant women have shown conclusive results. READ MORE
Former Glaxo Executive Indicted

November 10, 2010
A long-promised crackdown on pharmaceutical executives accused of potential wrongdoing is gaining momentum with the indictment yesterday of a former vice president and legal counsel of GlaxoSmithKline (Glaxo). While the pharmaceutical giant is headquartered in the UK, its US operations are based in North Carolina. READ MORE
READ MORE Birth Injury and Birth Defect Settlements and Legal News
READ MORE Drugs/Medical Settlements and Legal News
READ MORE Drugs/Medical Settlements and Legal News

READER COMMENTS
Shauna Staley
on
Joseph kelemen
on
Jes
on
South Carolina
on
West Virginia
on
Illinois
on
Puerto Rico
on
Texas
on
Wisconsin
on
Oklahoma
on
Texas
on
California
on
California
on
Oregon
on
Colorado
on
Maine
on
New Jersey
on
Colorado
on
Ohio
on
It is my understanding that when someone is in this �fight or flight� mode, their sense of judgment is broken down. Lack of judgment and compulsive behaviors ensued. I began leading a secret life and running up credit card debt. I had never exhibited this type of behavior before.
Not long after my doctor added another medication to my Wellbutrin (early June 2006) the compulsive tendencies began to wane. Shortly after that I stopped taking Wellbutrin.
I am still struggling with tens of thousands of dollars worth of debt resulting from this experience.
I have the records to show my medical history of taking the drug and the credit card records to show the debt that was incurred during that time.
The damage to my family (morally, financially, etc.) is done. I would like assistance in recovering whatever I can financially and can only hope that someone can help us to get the warning out so this doesn�t happen to anyone else!
Thank You!!
North Dakota
on
Minnesota
on
Texas
on
California
on
I am now employed at $52K annual but I strongly believe had I not been put on Wellbutrin as a deterrant to stop smoking I would not have suddenly quit my job and become so irrational. I was in Budapest on company business when I ran out of Wellbutrin on or around Nov 9, 2006. I did not know that stopping suddenly could throw me into such a tail spin. I had previously mentioned to the nurse at my DR's office that I some concerns a few weeks after originally being put on the substance. I was only asked if I stopped smoking while on it, and because I had she suggested I not stop taking it.
I believe it affected me for a long time after taking it. I moved from San Francisco to LA within a month of quitting and I did not want anything to do with people socially and I lost confidence in myself. I suffered so much stress that I began to get nose bleeds and I have continued to battle suicidal thoughts. I am in a business that is somewhat known and I was interviewed on a radio talk show in Feb 07 where they asked me why I suddenly quit. By then I had figured out that Wellbutrin had caused me to quit suddenly for seemingly no reason and I expressed that on the show.
I found this website by searching wellbutrin+suicide. I had no idea so many people have had the same or similar experience. I think it is a dangerous substance especially when given to people like me for the purpose of stopping smoking not depression. I fully support people being warned about this drug. If there is a class-action suit filed I would sign it.
I am a 41 year old male and I have been clean and sober for 18+ years in a 12 step program.