LAWSUITS NEWS & LEGAL INFORMATION
Enbrel
Patients who take Enbrel to treat certain types of arthritis may not be aware of the severity of Enbrel side effects they could be exposed to. These side effects include the risk of serious fungal infection and a risk of tuberculosis. Furthermore, the FDA is investigating a possible link between Enbrel and other drugs in its class, including Humira and Remicade, and the development of cancer in children and young adults.
FREE ENBREL SIDE EFFECTS LAWSUIT EVALUATION
Send your Enbrel Side Effects claim to a lawyer who will review your claim at NO COST or obligation.
GET LEGAL HELP NOW
GET LEGAL HELP NOW
Enbrel Side Effects
In November 2008, the maker of the Enbrel drug (known generically as etanercept) was required by the FDA to strengthen the drug's warnings about the risk of Enbrel side effects. Specifically, the label was updated to include a strengthened warning about the risk to some patients of developing opportunistic fungal infections while taking Enbrel.
 Although the warning label already included warnings about the risk of serious infections, the FDA noted in its Safety Alert that cases of histoplasmosis and other invasive fungal infections were not recognized consistently, resulting in a delay in treatment for the patient. According to an FDA news release (September 4, 2008), at least 21 reports of fungal infection were not treated immediately and, of those, 12 patients died.
Although the warning label already included warnings about the risk of serious infections, the FDA noted in its Safety Alert that cases of histoplasmosis and other invasive fungal infections were not recognized consistently, resulting in a delay in treatment for the patient. According to an FDA news release (September 4, 2008), at least 21 reports of fungal infection were not treated immediately and, of those, 12 patients died.
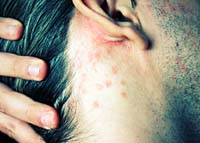
Symptoms of a systemic fungal infection include persistent fever, weight loss, sweats, cough, fatigue and shortness of breath. Patients who are taking Enbrel and notice these symptoms should seek medical attention because they could be experiencing serious side effects of Enbrel.
Enbrel is a tumor necrosis factor alpha blocker (TNF blocker) and is used to suppress the patient's immune system. It is used to treat moderate to severe rheumatoid arthritis, moderate to severe polyarticular juvenile idiopathic arthritis (formerly called juvenile rheumatoid arthritis), psoriatic arthritis, ankylosing spondylitis and moderate to severe plaque psoriasis.
The FDA had previously announced on May 1, 2008 that Enbrel's warning label was being updated to include a boxed warning alerting patients to the risk of infections including bacterial sepsis and tuberculosis.
Meanwhile, the FDA is investigating a possible association between the use of Enbrel and the development of lymphoma and other cancers in children and young adults. According to an FDA report (June 4, 2008), the agency is investigating approximately 30 reports over a 10-year period of cancer in children and young adults who used TNF blockers. Among the children and young adults who took Enbrel, cancers that were reported include lymphoma, leukemia, melanoma and solid organ cancers. The FDA has asked the makers of the TNF blockers to provide information about all cases of cancer in children who use those medications.
Enbrel is manufactured by Immunex Corporation and is marketed by Amgen and Wyeth Pharmaceuticals.
 Although the warning label already included warnings about the risk of serious infections, the FDA noted in its Safety Alert that cases of histoplasmosis and other invasive fungal infections were not recognized consistently, resulting in a delay in treatment for the patient. According to an FDA news release (September 4, 2008), at least 21 reports of fungal infection were not treated immediately and, of those, 12 patients died.
Although the warning label already included warnings about the risk of serious infections, the FDA noted in its Safety Alert that cases of histoplasmosis and other invasive fungal infections were not recognized consistently, resulting in a delay in treatment for the patient. According to an FDA news release (September 4, 2008), at least 21 reports of fungal infection were not treated immediately and, of those, 12 patients died.
Symptoms of a systemic fungal infection include persistent fever, weight loss, sweats, cough, fatigue and shortness of breath. Patients who are taking Enbrel and notice these symptoms should seek medical attention because they could be experiencing serious side effects of Enbrel.
Enbrel is a tumor necrosis factor alpha blocker (TNF blocker) and is used to suppress the patient's immune system. It is used to treat moderate to severe rheumatoid arthritis, moderate to severe polyarticular juvenile idiopathic arthritis (formerly called juvenile rheumatoid arthritis), psoriatic arthritis, ankylosing spondylitis and moderate to severe plaque psoriasis.
The FDA had previously announced on May 1, 2008 that Enbrel's warning label was being updated to include a boxed warning alerting patients to the risk of infections including bacterial sepsis and tuberculosis.
Meanwhile, the FDA is investigating a possible association between the use of Enbrel and the development of lymphoma and other cancers in children and young adults. According to an FDA report (June 4, 2008), the agency is investigating approximately 30 reports over a 10-year period of cancer in children and young adults who used TNF blockers. Among the children and young adults who took Enbrel, cancers that were reported include lymphoma, leukemia, melanoma and solid organ cancers. The FDA has asked the makers of the TNF blockers to provide information about all cases of cancer in children who use those medications.
Enbrel is manufactured by Immunex Corporation and is marketed by Amgen and Wyeth Pharmaceuticals.
Enbrel Legal Help
If you or a loved one has taken Enbrel and suffered from side effects, please click the link below to send your complaint to a lawyer who will evaluate your claim at no cost or obligation.Last updated on
ENBREL SIDE EFFECTS LEGAL ARTICLES AND INTERVIEWS
Enbrel Side Effects Could Include Infections
Virginia Man Files Lawsuit over Enbrel Side Effects

Fact or Coincidence? Enbrel Users Continue to Wonder

April 1, 2011
Enbrel side effects can have serious consequences on patients receiving the injection, including infections that affect the entire body or even tuberculosis, a severe infection of the lungs, according to the National Center for Biotechnology Information (NCBI). READ MORE
Virginia Man Files Lawsuit over Enbrel Side Effects

March 2, 2011
A Virginia man has filed a lawsuit claiming therapeutics company Amgen concealed Enbrel side effects, which allegedly led to his development of cancer, according to the Courthouse News Service. READ MORE
Fact or Coincidence? Enbrel Users Continue to Wonder

February 21, 2011
While Enbrel has been off the radar of late, the tumor necrosis factor alpha blocker (TNF blocker) is still on the minds of people who have used Enbrel to combat Enbrel arthritis or plaque psoriasis—and who have wound up with more than they had bargained for. READ MORE
READ MORE Drugs/Medical Settlements and Legal News

READER COMMENTS
Gerry Shea
on
Cassandra Cooper Brown
on
Jennifer A Leeds
on
Rita Smith
on
Marion Hackstedt
on
Teresa Markley
on
Sherri Zahand
on
Carla
on
Tristan
on
SAMUEL MONCADA
on
Kincaid
on
I take folic acid daily, women's vitamin, 1 predisone, and a probiotic. I'm active (Senior Games) and have a positive outlook and faith in God.
I guess it effects people differently, some people just rely on medicine, all of the warnings are on the boxes as well as inserts.
It's sad when lawyers as well as the individuals themselves try to make money off of medicines and prey on people with illnesses.
Sylvia Shavings
on
Jessica Sanchez
on
Helen Scow
on
Jodau
on
ella patten
on
Catherine
on
Linda Morton
on
alan schlossberg
on
i first developed a serious fungal infection in my mouth nose & throat & was unable to eat or drink & became de hydrated
This then followed shingles in my left ear,that became very painful,until I became unconscious & was hospitalized for 2 weeks.
When i finally woke up my face was twisted & distorted,& i was unable to open or close my left eye,or hear from my left ear
I could not walk because my balance was off
After many different tests I was advised that I had Ramsey Hunt Desease that destroyed the 7th & 8th cranial nerves & that there was no cure for this .
I have had 2 surgeries to my left eye to enable me to open & close it,& use drops 6 times daily in that eye to protect the cornea for the rest of my life.
My balance & hearing continue to be a problem,however the worst part is the dryness i experience in sinuses & throat which interferes with my sleep & digestion,as a result of limited saliva.
This all could have been prevented if either of the two doctors who had been treating me on a regular basis would have recognized these side effects,& given me an anti virus.
It would have save me from this horrible desease
Cheryle Rolnick
on
Jim Lewandowski
on
Florida
on
Anonymous
on
My heart doctor said I had a minor heart attack. I told the prescribing doctor and she was surprised. I never tried it again.
Georgia
on
I have also been hospitalized after starting Humira in 2007 for Acemic Colitis and the flare ups continue to the present day. I have also been diagnosed with a fungul infection that has to date gone untreated.
California
on
Massachusetts
on
West Virginia
on
Missouri
on
South Carolina
on
Ontario
on
Ohio
on
Indiana
on
Ohio
on
Florida
on