LAWSUITS NEWS & LEGAL INFORMATION
Stevens Johnson Syndrome Linked to Bactrim
Bactrim (sulfamethoxazole and trimethoprim) is an antibiotic prescribed to adults and children to treat ear infections, bronchitis, pneumonia, urinary tract infections and other bacterial infections. However, bactrim side effects are numerous. —The most common and potentially lethal side effect is Stevens Johnson Syndrome (SJS)and toxic epidermal necrolysis (TEN).
 Bactrim was approved by the FDA in 1973. Since that time, Bactrim and the combined antibiotic treatment of Bactrim and Septra (known as trimethoprim-sulfamethoxazole) have caused severe allergic reactions in countless patients, including Stevens-Johnson Syndrome (SJS) and Toxic Epidermal Necrolysis (TENS).
Bactrim was approved by the FDA in 1973. Since that time, Bactrim and the combined antibiotic treatment of Bactrim and Septra (known as trimethoprim-sulfamethoxazole) have caused severe allergic reactions in countless patients, including Stevens-Johnson Syndrome (SJS) and Toxic Epidermal Necrolysis (TENS).
Stevens Johnson Syndrome
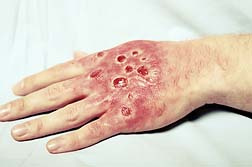
Stevens Johnson Syndrome (SJS) is a rare inflammatory skin disorder that can be life-threatening. The disease causes lesions in the mouth, eyes and on the body. Stevens Johnson Syndrome can occur at any age although the majority of cases range between the ages of 20 and 40. One-fifth of the cases occur in people under the age of 20.
Symptoms of SJS include: fever, sore throat, and headache, difficulty breathing, inflammation/swelling of throat, lips, tongue and face, hives, skin lesions, blisters, and unusual bleeding.
In some cases, permanent skin damage and scarring occur. Lesions on internal organs can cause serious inflammation of the lungs, heart and kidneys. Up to 27 percent of those affected by Stevens Johnson Syndrome or related illnesses incur long term eye damage or vision loss. Upwards of 15 percent of patients with this syndrome die from the condition.
SJS can become deadly and requires medical attention as soon as allergic reactions are noticeable.
Those with kidney or liver disease are not advised to take Bactrim; the drug is not approved for infants under 2 months old and it is classified by the FDA as category C, since the effects of Bactrim on a fetus are unknown.
Last updated on
FREE BACTRIM SJS LAWSUIT EVALUATION
Send your Bactrim SJS claim to a lawyer who will review your claim at NO COST or obligation.
GET LEGAL HELP NOW
GET LEGAL HELP NOW
Bactrim Stevens Johnson Syndrome
 Bactrim was approved by the FDA in 1973. Since that time, Bactrim and the combined antibiotic treatment of Bactrim and Septra (known as trimethoprim-sulfamethoxazole) have caused severe allergic reactions in countless patients, including Stevens-Johnson Syndrome (SJS) and Toxic Epidermal Necrolysis (TENS).
Bactrim was approved by the FDA in 1973. Since that time, Bactrim and the combined antibiotic treatment of Bactrim and Septra (known as trimethoprim-sulfamethoxazole) have caused severe allergic reactions in countless patients, including Stevens-Johnson Syndrome (SJS) and Toxic Epidermal Necrolysis (TENS). Stevens Johnson Syndrome
Stevens Johnson Syndrome (SJS) is a rare inflammatory skin disorder that can be life-threatening. The disease causes lesions in the mouth, eyes and on the body. Stevens Johnson Syndrome can occur at any age although the majority of cases range between the ages of 20 and 40. One-fifth of the cases occur in people under the age of 20.
Symptoms of SJS include: fever, sore throat, and headache, difficulty breathing, inflammation/swelling of throat, lips, tongue and face, hives, skin lesions, blisters, and unusual bleeding.
In some cases, permanent skin damage and scarring occur. Lesions on internal organs can cause serious inflammation of the lungs, heart and kidneys. Up to 27 percent of those affected by Stevens Johnson Syndrome or related illnesses incur long term eye damage or vision loss. Upwards of 15 percent of patients with this syndrome die from the condition.
SJS can become deadly and requires medical attention as soon as allergic reactions are noticeable.
Those with kidney or liver disease are not advised to take Bactrim; the drug is not approved for infants under 2 months old and it is classified by the FDA as category C, since the effects of Bactrim on a fetus are unknown.
Bactrim SJS Legal Help
If you or a loved one has suffered from Stevens Johnson Syndrome after taking Bactrim, please click the link below to send your complaint to a lawyer to evaluate your claim at no cost or obligation.Last updated on
BACTRIM SJS LEGAL ARTICLES AND INTERVIEWS
Woman's Skin Melts Off From Stevens-Johnson Syndrome
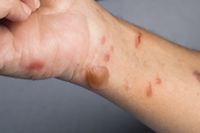
Stevens Johnson Syndrome Victim Has Permanent Scarring in her Mouth
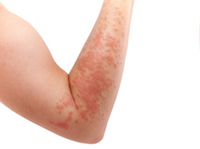
Texas Woman Suffers Stevens Johnson Syndrome from Bipolar Medication


May 13, 2017
Snellville, GA: A Georgia woman claims that her skin melted off when she contracted Stevens-Johnson Syndrome (SJS) after being given an incorrect dosage of an antidepressant by a pharmacist. READ MORE
Stevens Johnson Syndrome Victim Has Permanent Scarring in her Mouth

April 17, 2017
Tampa, FL: Donna Rushing is a resident from Tampa who developed Stevens Johnson Syndrome from a medication she had been taking. While she was spared hospitalization and more serious SJS side effects that can often include massive loss of skin and life-threatening infections, her battle with Stevens Johnson skin disease was enough to cause her to lose weight, affected her eating and has left her with permanent scarring in her mouth. READ MORE
Texas Woman Suffers Stevens Johnson Syndrome from Bipolar Medication

March 19, 2017
Houston, TX: The majority of patients and plaintiffs touched by the scourge of Stevens Johnson Syndrome (SJS) can usually track, or at the very least suspect with a reasonable hypothesis that Stevens Johnson Syndrome symptoms materialize through the use of pharmaceutical drugs and medications including standard, everyday products such as over-the-counter (OTC) pain medication. READ MORE
READ MORE Stevens Johnson Syndrome (SJS) Settlements and Legal News
READ MORE Drugs/Medical Settlements and Legal News
READ MORE Drugs/Medical Settlements and Legal News

READER COMMENTS
Lisa Dyess
on
Ken King
on
Nothing was different during that time, except taking Bactrim. They did a Biopsy on my lung. Now I have Cancer there. The dose was 800mg./160mg. Twice per day. Dr. Said stop taking after Biopsy came back. I think the Bactrim caused my Cancer.
Theresa
on
Leonor sanchez
on
Stacy
on
Joe in virginia
on
Kayla
on
amber meltzer
on
Anonymous
on
Virginia
on
North Carolina
on
Oregon
on
Alabama
on
North Carolina
on
Florida
on
I spoke with Winn-Dixie pharmacy public relations several times, faxed to their corporate HQ here in Jax, Fl requesting they amend their drug information sheet to include these symptoms. They should immediately discontinue this med, but not one change have they made. I am so disappointed. This information should be in all drug information sheets.