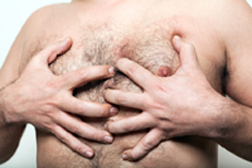
 In 2003, when Nicholas Murray was nine years old, he was prescribed Risperdal to treat schizophrenia, but the antipsychotic drug had only been approved by the FDA for adults. The Maryland boy developed gynecomastia, the development of female-like breasts in boys and young men, and the jury found Risperdal to be a “substantial factor” in this disorder. The jury found the drug company guilty of failing to warn patients of the risks of gynecomastia in connection with the drug.
In 2003, when Nicholas Murray was nine years old, he was prescribed Risperdal to treat schizophrenia, but the antipsychotic drug had only been approved by the FDA for adults. The Maryland boy developed gynecomastia, the development of female-like breasts in boys and young men, and the jury found Risperdal to be a “substantial factor” in this disorder. The jury found the drug company guilty of failing to warn patients of the risks of gynecomastia in connection with the drug.The first Risperdal trial in February 2015 awarded plaintiff Austin Pledger and his family $2.5 million. Pledger, who is now 20, was prescribed Risperdal when he was eight years old to treat symptoms of autism. In his teenage years, Pledger developed size 46 DD breasts. Risperdal lawsuits also allege that the drug company encouraged off-label prescriptions for children before the breast growth side effect went on the label. At the trial, Pledger’s attorney said “If there was ever a clear failure-to-warn case, this was it.”
According to court documents, J&J encouraged doctors to prescribe Risperdal off-label to treat attention deficit and disruptive behavior and other childhood disorders, which are, for the most part, simply symptoms of growing up.
READ MORE RISPERDAL LEGAL NEWS
Kessler also added that J&J promoted Risperdal off-label, going so far as to provide doctors with trinkets such as children’s bright-colored Lego-like blocks featuring the Risperdal logo.
The third case found in favor of the defense, Janssen Pharmaceuticals, rather than plaintiff William Cirba. Although the jury determined that J&J failed to warn of gynecomastia risks, no damages were awarded because a direct link between Risperdal and Cirba’s breast growth was not established.
More than 1,500 Risperdal lawsuits nationwide are waiting to go to trial.

READER COMMENTS
Michele Mitchell
on
Kimberly Hughes
on
Nereida Guillén
on