 The jury in the first of hundreds of trials consolidated in US District Court Minneapolis before Judge John Tenheim, returned its verdict earlier this month, according to the December 7 issue of the Minneapolis Star Tribune. Total Levaquin claims overall in federal and state courts number in the thousands. It is not known if the defendants will appeal. However, the victory does bode well for other victims of Levaquin tendon ruptures.
The jury in the first of hundreds of trials consolidated in US District Court Minneapolis before Judge John Tenheim, returned its verdict earlier this month, according to the December 7 issue of the Minneapolis Star Tribune. Total Levaquin claims overall in federal and state courts number in the thousands. It is not known if the defendants will appeal. However, the victory does bode well for other victims of Levaquin tendon ruptures.The impact on the life of the plaintiff is compelling. John Schedin is now 82, but he was a spry 76 when he went to his doctor complaining of a severe chest cold. His doctor prescribed Levaquin plus a steroid in an effort to knock the infection down.
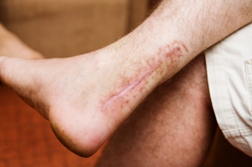
However, the plaintiff suffered a rupture to both of his Achilles tendons after only three days on the drug. In court testimony, the doctor who prescribed the Levaquin medication to Schedin noted that while he was aware of the risk for tendon problems, he was not aware that Levaquin taken in concert with a steroid could dramatically increase the risk for tendon rupture in seniors.
As a result of his Levaquin tendon ruptures, Schedin has been reduced to crawling up the stairs on his hands and knees for the journey to his bed at night. He testified at trial that he can no longer vacation with his family and that his quality of life has been negatively impacted.
Defense witnesses testified that Schedin's injury might have been complicated by high blood pressure, high cholesterol levels and the steroid he had been prescribed.
READ MORE LEVAQUIN LEGAL NEWS
Schedin's prescribing doctor testified that he did not recall any sales rep from Johnson & Johnson and/or Ortho-McNeil-Janssen making reference to tendon risk with Levaquin during sales calls.
The manufacturer stands behind its product, and said through a lawyer that no information has ever been withheld or concealed. Schedin was awarded $1.1 million in punitive damages and $700,000 in compensatory damages. Levaquin has consistently earned annual sales approaching the billion-dollar level since its US introduction in 1997, and was the most-prescribed antibiotic in the world by 2006.

READER COMMENTS
ralph a. applegate
on
David Lee Boswell
on