 That hasn’t stopped manufacturers from expanding their respective empires.
That hasn’t stopped manufacturers from expanding their respective empires.Zimmer Holdings (Zimmer), for example, is the manufacturer of the ill-fated Zimmer Durom Cup. According to Business Monitor Online (2/4/15), Zimmer reported fourth-quarter losses and a slump for 2014 overall compared with the previous year. Revenue was reported to be higher last year than in 2013, with a 1.1 percent increase overall. But the good news on the revenue front was offset by special charges related to many a Hip Replacement Lawsuit, and the acquisition of Biomet, amongst other costs.
That said, the integration of Biomet is said to be important to Zimmer’s future, in spite of the cost.
Meanwhile, over at Stryker Corporation, the acquisition of OtisMed has not come without a cost. To that end, Stryker and OtisMed were required to pay almost $80 million to settle a criminal complaint issued by the US Department of Justice (DOJ). The charges related to the sale and distribution of medical devices by OtisMed that occurred without regulatory approval.
The DOJ duly noted that OtisMed’s criminal activity occurred before the acquisition by Stryker Corp., and also occurred without Stryker’s “prior knowledge or acquiescence.” Stryker Corp, the manufacturer of the problematic Rejuvenate Modular Hip System, was caught up in the issue nonetheless.
OtisMed, the Kalamazoo Gazette (12/14/14) reports, marketed what was known as the OtisKnee cutting guide, a tool to assist surgeons in making more accurate bone cuts during knee replacement procedures. According to the DOJ, OtisMed featured the OtisKnee in promotional and marketing materials before the device was properly evaluated by the US Food and Drug Administration (FDA). The device, in the end, did not receive clearance from the FDA, which rejected OtisMed’s application. That didn’t stop OtisMed from selling 18,000 of the devices between May 2006 and September 2009, generating $27.1 million in revenue in the process.
This all occurred prior to September 2009. Stryker purchased OtisMed two months later, in November of that year, and thus was not involved in the apparent skullduggery. OtisMed was subsequently fined $34.4 million, as well as the forfeiture of $5.16 million as sentencing in the criminal case. Stryker also entered into a settlement with the DOJ, but the details were not disclosed and Stryker made no comment.
Combined, the total payout was $79,560,400 plus interest to settle the complaint.
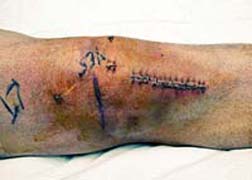
It was an expensive fall for Stryker. In addition to the settlement, Stryker announced a $1.4 billion fund (at minimum) to settle a host of Hip Replacement lawsuits following Hip Replacement Implant Failure associated with one of its product lines, which it recalled in 2012. While the Rejuvenate Modular Hip System was not a true metal-on-metal device, the product did feature some metal components that proved problematic for scores of patients.
READ MORE HIP AND KNEE REPLACEMENT LEGAL NEWS
This has resulted, for many Americans, in the need for revision surgery to replace the problematic joint. Revision procedures are often more complex than the initial surgery, with longer healing times. The negative impact on one’s health, livelihood and career has left many a plaintiff running to their Hip Replacement Lawyer or filing a Knee Replacement lawsuit.

READER COMMENTS
Michael Harris
on