 The lawsuits all name Fresenius Medical Care Holdings Inc., together with a number of other business entities within the Fresenius banner. The lawsuits are similar in nature to thousands of lawsuits that have surfaced since a GranuFlo recall - and a similar recall involving NaturaLyte - occurred in the spring of 2012.
The lawsuits all name Fresenius Medical Care Holdings Inc., together with a number of other business entities within the Fresenius banner. The lawsuits are similar in nature to thousands of lawsuits that have surfaced since a GranuFlo recall - and a similar recall involving NaturaLyte - occurred in the spring of 2012.It should be noted that the recall, undertaken voluntarily by Fresenius and approved by the US Food and Drug Administration (FDA), did not involve recalling actual product - nor was there any suggestion of problems with the products. Rather, the recall involved product labeling.
Nonetheless, the issue surrounding the labels for GranuFlo and NaturaLyte allegedly led to serious Alkali Dosing Errors that negatively impacted the health of a countless number of dialysis patients.
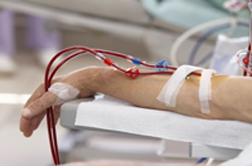
GranuFlo and NaturaLyte are two products used in the dialysis process. Both are manufactured by Fresenius, a manufacturer of dialysis products as well as an operator of dialysis clinics based in Waltham, Massachusetts. NaturaLyte is shipped in liquid form, while GranuFlo is a concentrate shipped as a dry powder, and requires mixing to the proper concentration by the end user. Integral to a successful dialysis is the careful monitoring of bicarbonate levels in the blood - levels that can reportedly spike if the absolute correct levels and volumes for GranuFlo and NaturaLyte are not achieved.
Such alkali dosing errors can elevate bicarbonate in the blood, which can then risk cardiovascular issues such as a GranuFlo heart attack.
An issue amongst the thousands of lawsuits already in the pipeline is the circumstances surrounding an internal memo that Fresenius circulated within its own dialysis clinics pertaining to potential issues with GranuFlo and NaturaLyte, and directing Fresenius doctors and medical support staff to monitor dialysis patients carefully.
That memo was not circulated to other dialysis clinics outside of the Fresenius umbrella but also using GranuFlo and NaturaLyte, until the memo was leaked to the FDA in early 2012, some weeks after the original memo was issued. An FDA investigation prompted Fresenius to issue a voluntary Fresenius Medical Care recall.
Plaintiffs claim the recall - which again affected product labels and not the product itself - was too little, too late. Plaintiffs involved with a DaVita lawsuit hold that a DaVita Heart Attack occurring from use of GranuFlo and NaturaLyte by the rival to Fresenius may have been prevented if DaVita had been informed about the potential for Alkali Dosing Errors at the same time as Fresenius dialysis clinics were notified.
The Kentucky lawsuits allege that Fresenius might have been aware of a potential issue as early as 2001. An internal communication dated March 2001 and sent to medical directors and admitting physicians at Fresenius treatment centers addressed a preliminary analysis concerning certain patient scenarios and risks for adverse effects, including cardiopulmonary arrest (CPA).
Fresenius spokesperson Kent Jarrell, in comments to the Glasgow Daily Times (10/24/14), a newspaper based in Kentucky, defends the communication, noting that the issue was preliminary at best. “In this preliminary analysis, no conclusion was suggested or made that the product caused the CPA in the patients,” Jarrell said in comments published in the Glasgow Daily Times. “Our subsequent reviews of specific patients suggest that other factors unique to each patient caused the CPA. Dialysis patients with advanced and permanent kidney failure often have other serious illnesses, including heart or lung disease. And since the November 2011 preliminary analysis, two published studies found no meaningful association between [those scenarios] and mortality.”
READ MORE GRANUFLO RECALL LEGAL NEWS
As of November 17, there were no fewer than 2,220 claims pending in federal multidistrict litigation (In re: FreseniusGranuFlo/NaturaLyte Dialysate Litigation, MDL No. 2428, US District Court, District of Massachusetts). Another 3,900 claims are pending at the state level at Massachusetts’ Middlesex County Superior Court (In re: Consolidated Fresenius Cases, No. MICV2013-03400-O).
A Fresenius spokesperson noted that any lawsuit stemming from NaturaLyte Liquid Acid Concentrate recall or NaturaLyte GranuFlo recall is without merit.

READER COMMENTS
Cathy veniero
on