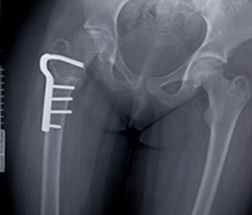
 According to The Star-Ledger of Newark, New Jersey (4/10/13), it was Glynn’s femur that let go that April day in 2009, when the Cohoes, New York resident was 54. Fosamax side effects include a higher incidence of femur fracture, which is ironic considering that Fosamax is prescribed as a preventative strike against osteoporosis and is designed to actually strengthen bone.
According to The Star-Ledger of Newark, New Jersey (4/10/13), it was Glynn’s femur that let go that April day in 2009, when the Cohoes, New York resident was 54. Fosamax side effects include a higher incidence of femur fracture, which is ironic considering that Fosamax is prescribed as a preventative strike against osteoporosis and is designed to actually strengthen bone.However, in the process, there appears to be a link between Fosamax and low-impact fractures of the femur. Glynn, her Fosamax lawsuit claimed, had simply bent over to pick up an object when she experienced a spontaneous fracture of her leg.
As it happened, according to Law 360 (6/27/13), the jury found in favor of Fosamax manufacturer Merck & Co. Given that the ruling in the Fosamax femur lawsuit went against the plaintiff, there was no ruling on liability (Glynn v. Merck Sharp & Dohme Corp., case number 3:11-cv-05304, in the U.S. District Court for the District of New Jersey).
However, Merck had submitted for summary judgment and the judge heard arguments on the motion prior to the jury ruling, but waited until after the jury verdict to deliver his ruling on summary judgment. The decision provides some insight into the federal regulator, the US Food and Drug Administration (FDA), that approved Fosamax in the first place.
According to Law 360, US District Court Judge Joel A. Pisano noted that a big part of Glynn’s case was an accusation of failure to warn on the part of Merck, the manufacturer of Fosamax. However, Merck had actually recommended to the FDA in 2009 that the prescribing label for Fosamax be updated to include warnings for potential Fosamax femur fractures.
Incredibly, the FDA said no.
As outlined in Judge Pisano’s opinion, the FDA in 2008 asked Merck to investigate a possible link between the use of bisphosphonates (Fosamax) and the emergence of atypical fractures to the femur that are low-impact and non-traumatic. Merck looked into it and concluded there was, indeed, a potential link between bisphosphonate use and stress fractures - and recommended to the FDA in 2009 that labeling for Fosamax be updated in kind.
However, the FDA refused, citing insufficient evidence. It would be another two years before the FDA made the label change, long after Glynn filed her Fosamax lawsuit in 2009. Thus, the judge granted summary judgment to Merck over the failure to warn argument given that Merck, it appears, tried in vain to do just that - but was rebuffed by the very agency intended to protect the public health.
As for Merck, according to transcripts of Glynn’s trial, the manufacturer may have known five years before Fosamax arrived on the market that there was a problem with bisphosphonates, according to a published report in The Star-Ledger. Glynn’s Fosamax attorney, at the start of her trial, noted that consultants began sounding the alarm bells to Merck as early as 1990 that Fosamax and femur fractures could be a problem. The reason, the consultants were saying, was that bisphosphonate use in some patients appeared to hamper the bone’s natural capacity to self-repair and heal micro-fractures. Such a collection of micro-fractures, left unchecked due to the use of bisphosphonates such as Fosamax, could lead to such spontaneous fractures of the femur.
READ MORE FOSAMAX LEGAL NEWS
Was Glynn caught in the crosswind of inactivity by both entities? And how many consumers may have been harmed in the 21 years between the first claimed reports of spontaneous femur fractures by consultants starting in 1990, and the FDA-mandated label change in 2011? Thousands of pending Fosamax femur fracture lawsuits may be the answer to that question…

READER COMMENTS
ANONYMOUS
on