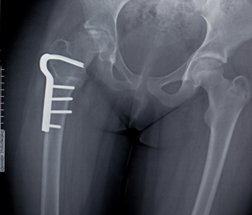
 According to The New York Times (09/05/11), two FDA advisory panels and FDA staff will meet on September 9 to examine the use of Fosamax and other bisphosphonates. Specifically, they will consider whether women who use bisphosphonates should be required to take periodic breaks from the medications because of the risk of rare, but serious side effects. The panels will also reportedly review medical data to determine whether bisphosphonates are safe and effective when used for longer than three to five years.
According to The New York Times (09/05/11), two FDA advisory panels and FDA staff will meet on September 9 to examine the use of Fosamax and other bisphosphonates. Specifically, they will consider whether women who use bisphosphonates should be required to take periodic breaks from the medications because of the risk of rare, but serious side effects. The panels will also reportedly review medical data to determine whether bisphosphonates are safe and effective when used for longer than three to five years.Studies have linked Fosamax to an increased risk of femur fractures, osteonecrosis of the jaw (jawbone death) and esophageal cancer. Bisphosphonates are prescribed to prevent a loss of bone mass, and have been shown to reduce fractures of the hip and spine in some women.
Although research on bisphosphonates tends to cover a three to five year period, many women take the drugs longer than that to prevent osteoporosis. The risks of bisphosphonates, however, have been shown to increase with longer use.
In a report released prior to the FDA's advisory panel meeting (09/09/11), the FDA noted that results from some studies "suggest no significant advantage of continuing drug therapy beyond 5 years." The report further states that the safety of long-term use of bisphosphonates is not clear because of conflicting results from studies.
READ MORE FOSAMAX LEGAL NEWS
The FDA noted that it "has not concluded that taking an oral bisphosphonate drug increases the risk of esophageal cancer," (07/21/11).
At this point, the FDA advises that the benefits of oral bisphosphonates outweigh the risks.
Meanwhile, Merck, maker of Fosamax, faces lawsuits alleging the medication causes the development of osteonecrosis of the jaw. One lawsuit, filed by Linda Secrest, will begin jury selection around the time the FDA issues its report. Although Merck has won three lawsuits alleging Fosamax caused jawbone death, the company did lose one case, in which a jury awarded the plaintiff $8 million, according to Bloomberg News (06/25/10).

READER COMMENTS
Susan Linton
on