Fosamax, manufactured for over a decade by Merck, has been doing quite well with its osteoporosis drug, thank you. In fact, in spite of a basket of other drugs available designed to fight bone loss, WHEC Television in Rochester, New York reported that more than half of all women in the United States suffering from osteoporosis take Fosamax. Other sources suggest that more than 10 million men and women have taken Fosamax.
 This number is bound to increase, now that the patent has expired. Merck no longer has exclusivity. Thus, there will be new, and most likely cheaper versions of Fosamax out there.
This number is bound to increase, now that the patent has expired. Merck no longer has exclusivity. Thus, there will be new, and most likely cheaper versions of Fosamax out there.What hasn't changed is the formulation. And that could be a problem, given the number of patients having experienced, and are suing Merck over the development of osteonecrosis, or bone death in the jawbone.
There are other side effects as well, which appear to be the basis of a lawsuit launched against Merck by a Connecticut woman over claims that Fosamax caused serious side effects. JoAnn Moranski of Fairfield says she has taken Fosamax since it first came onto the market in 1996, and claims that Fosamax caused stress fractures and suppressed bone regeneration in her legs. She stopped taking the drug in 2006.
The Wall Street Journal reported on January 28th that more than 400 people have already sued Merck, alleging that Fosamax causes the bone-wasting osteonecrosis, or ONJ, of the jaw.
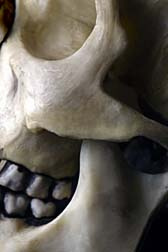
ONJ is a nasty affliction. Osteonecrosis of the jaw causes severe infections, swelling and loosening of teeth. Also known as dead-jaw syndrome, ONJ is a disease in which bone tissue in the jaw does not heal after minor traumas. Dental extractions that cause bone to become exposed can lead to fractures and infections often requiring long-term antibiotic therapy and surgery to remove the dead and dying bone tissue.
Most cases of ONJ seem to stem from the use of IV bisphosphonates, which are administered intravenously and are the more potent form of alendronate.
Judith, of Leavenworth KS didn't need the potent IV version of Fosamax to have issues. Although she didn't suffer from the debilitating ONJ, the retired teacher tells Consumersaffairs.com that she took Fosamax tablets for eight years to combat osteoporosis.
All appeared well until she started experiencing pain in her left leg. A neurologist prescribed various meds, physical therapy and acupuncture. Judith writes that she was also given, at a pain clinic, steroid shots in the groin and spine.
She could find no relief until a friend tipped her off about Fosamax. Judith stopped taking Fosamax on September 1st of last year. After a month she found that she could sleep at night without pain. By November, she could navigate stairs without pain.
That latter improvement was bittersweet for Judith, who was forced to sell her beautiful two-story home because she was in too much pain to navigate the stairs. The master bedroom was on the upper floor.
Now, with the pain gone, it appears that Fosamax was the culprit, and it was all for naught. What's more, Judith writes that she had to retire early from her teaching position, and had to pay out-of-pocket for expensive steroid shots that set her back $5,000 alone. Various other meds, therapies and treatments only increased that cost.
And the ultimate irony: after eight years on a medication that was supposed to diminish the effects of osteoporosis, she has found that her condition has worsened, not improved.
Now, it appears, even more Americans have the potential to benefit from generic, and much-less-expensive versions of Fosamax. It has been reported that Merck earned $3 billion from Fosamax sales in 2007. That number is surely to drop now that the company has lost patent protection, and there will be new versions of the drug available.
While that may be good news for health care plans, and anyone who found the cost of Fosamax prohibitive, the downside is that a drug with apparent issues will now be readily available to more patients, thanks to the cost reduction that is almost always delivered by generics.
It is interesting to note that as part of the February 6th 2008 communiqué announcing the licensing of the first (of probably many) Fosamax generic to Teva Pharmaceuticals USA of North Wales, PA, the FDA stated:
"Generic drug manufacturers must demonstrate that a generic drug has the same active ingredient, dosage form, strength, route of administration, quality and performance characteristics, among other things, as the approved brand-name drug."
And...
"The FDA works to assure the safety and efficacy of generic drugs through a rigorous scientific and regulatory process," said Gary J. Buehler, R.Ph, director of the FDA's Office of Generic Drugs. "These approvals will provide generic options for patients who take Fosamax for their osteoporosis."
READ MORE LEGAL NEWS
Over to you, Judith:
"In discussing this with my doctor, last week, she said that the FDA has recently notified the doctors of this adverse side effect from Fosamax. I want to check and see if others have sought legal action because of the muscle and joint pain caused by Fosamax."
In spite of approving a generic version of Fosamax mere days after Merck's patent expired, the FDA is continuing its safety review of alendronate, and bisphosphonates.
