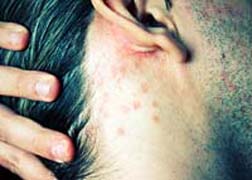
 Blogger 'jennybyc' wrote, "not only did it not make me any better, it made me worse. My fatigue was terrible and I developed swelling under my skin that almost looked like huge fluid-filled cysts. My doc had never seen such a thing."
Blogger 'jennybyc' wrote, "not only did it not make me any better, it made me worse. My fatigue was terrible and I developed swelling under my skin that almost looked like huge fluid-filled cysts. My doc had never seen such a thing."While not identifying the reason for her Enbrel prescription, 'jennybyc' goes on to say that after being off Enbrel for two years most of the swelling originally caused by the Enbrel allergic reaction had subsided, save for "a couple of patches on my arms that I don't think will ever go away."
Meanwhile, a new study has found that use of TNF blockers in children may increase the risk of malignancy, with the indication for inflammatory bowel disease identified as a particular concern, according to MedPage Today back in July. Additionally, the publication Arthritis and Rheumatism reported in August the findings of Peter Diak, PharmD and colleagues from the US Food and Drug Administration (FDA) that a total of 48 cases of malignancy had been reported among children and young adults taking TNF blockers—Enbrel among them.
Sourcing the FDA's Adverse Reporting System database, the researchers found that among the 9,200 children treated with etanercept (Enbrel), there were 15 reported malignancies with five cases of lymphoma, four of leukemia, two cases each of malignant melanoma and thyroid cancer, and one case each of myelodysplasia, yolk sac tumor and bladder cancer.
Mike S. blogged about his concern that Enbrel may have had a part to play in a diagnosis of acute lymphoblastic leukemia. The 28-year-old had been taking Enbrel for two years to control psoriasis when his diagnosis occurred.
The FDA released a communiqué in August of 2009 with regard to the potential for tumor necrosis factor alpha-blockers such as Enbrel to be associated with an increased risk of malignancies in children and adolescents. While Mike was hardly an adolescent at 28 when he blogged about his condition not long after the FDA advisory was released, he remained concerned nonetheless that in his view Enbrel side effects may have had a part to play in his diagnosis.
Enbrel was among the TNF blockers that came under FDA scrutiny starting in June of 2008. That review led to an advisory a little more than a year later, coupled with a requirement for TNF blocker manufacturers to include cancer warnings on product labeling.
READ MORE ENBREL SIDE EFFECTS LEGAL NEWS
As for Mike S., did Enbrel reaction trigger his leukemia? At the time of his posting, his leukemia was in remission. Blogger Ed Dewke, who had a testicle removed due to the presence of a cancerous tumor, had been taking Enbrel for psoriasis when he noted the sudden onset of massive swelling. Was the use of Enbrel and his tumor related? Dewke has ruled that out. But the coincidence remains.

READER COMMENTS
Noel
on
Theodore john Kountouris
on
Theodore john Kountouris
on
MICHAEL STOREMSKI
on
Mike S.
on
Mike S.
on
Please, anyone on enbrel or considering it, monitor your bloodwork and CBC's VIGILANTLY! Consider using something less dangerous and more understood. Who knows what side effects could pop up in 20 years or more from these biologics.
I also find it ironic that I took a drug to control my psoriasis, that in my opinion, played a part in causing leukemia, and the big pharm company behind it profited nicely.
Then the same big pharm company profited again when I took a drug I needed to stimulate my white blood cell counts after chemotherapy doses.
They get you sick and profit off you, then make you better and profit off you again.
Glenn bloomer
on