 That strategy affects all extended-release and long-acting opioid medications, which are used to treat moderate and severe pain. Included in that are Duragesic pain patches (Duragesic is known generically as fentanyl), OxyContin and Avinza. According to the FDA, these drugs are often misprescribed, misused and abused, putting patients at risk of overdose and, in some cases, death.
That strategy affects all extended-release and long-acting opioid medications, which are used to treat moderate and severe pain. Included in that are Duragesic pain patches (Duragesic is known generically as fentanyl), OxyContin and Avinza. According to the FDA, these drugs are often misprescribed, misused and abused, putting patients at risk of overdose and, in some cases, death.The new REMS focuses on educating doctors about pain management and patient selection to ensure the drugs are only prescribed to patients who meet criteria for opioid treatment. Furthermore, companies that manufacture the pain relievers are being asked to include a medication guide that explains safe use of the drugs. Manufacturers must propose their REMS plans within 120 days of the FDA's April 19 announcement.
In making its announcement, the FDA noted that more than 33 million Americans aged 12 and older misused opioid medications in 2007.
A Risk Evaluation and Mitigation Strategy is a plan developed and implemented by a drug company—at the request of the FDA—to manage serious risks associated with a medication. The strategy is used to ensure that a drug is used properly and that the drug's benefits still outweigh the risks.
READ MORE DURAGESIC FENTANYL PATCH LEGAL NEWS
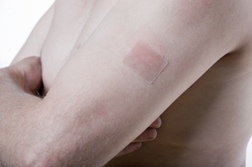
Duragesic pain patches are different from other opioids in that they are delivered via a transdermal pain patch—a patch that is placed on the skin. Fentanyl pain patches have been recalled numerous times because of issues with manufacturing that have resulted in patients receiving much more of the active ingredient (fentanyl) at once than intended. Fentanyl patches are recommended only for patients who have chronic, debilitating pain that is not controlled with other painkillers and who have a tolerance for opioid medications. Lawsuits against the makers of fentanyl patches allege that improper manufacturing resulted in the accidental deaths of some fentanyl patients.

READER COMMENTS
Rhonda Davis
on