 For women with epilepsy, taking Depakote was an absolute necessity. After all, there are serious risks to having seizures and Depakote provides important treatment. But some women were prescribed Depakote for migraines--painful, for sure, but some women say not worth the risks to their unborn babies. Furthermore, there may be other drugs available to treat seizures and migraines that do not carry the same risks as Depakote (known generically as valproic acid).
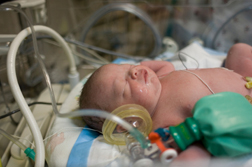
For women with epilepsy, taking Depakote was an absolute necessity. After all, there are serious risks to having seizures and Depakote provides important treatment. But some women were prescribed Depakote for migraines--painful, for sure, but some women say not worth the risks to their unborn babies. Furthermore, there may be other drugs available to treat seizures and migraines that do not carry the same risks as Depakote (known generically as valproic acid).In June 2011, the US Food and Drug Administration (FDA) issued an update about valproate and related products, warning that children exposed to the drugs prior to birth tended to have lower IQ and other cognitive test scores than children exposed to other anti-seizure medications prior to birth. Even before this time, valproate drugs carried a warning about the risk of serious neural tube defects and other birth defects associated with use of the drug while pregnant.
READ MORE DEPAKOTE BIRTH DEFECT LEGAL NEWS
Among the birth defects that the FDA has issued warnings or reminders about are neural tube defects (including spina bifida), craniofacial defects and cardiovascular defects. According to the FDA, the risk of a neural tube defect in the general population is 1 in 1,500. In babies born to mothers who used valproate during the first twelve weeks of pregnancy, that risk increases to 1 in 20 babies.
