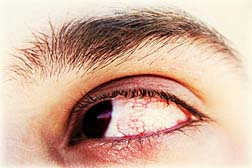
According to the Centers for Disease Control and Prevention, at least four patients have developed an Acanthamoeba keratitis infection since the AMO Complete recall was issued on May 25. The agency says that many users were unaware that the contact lens solution had been recalled.
 In spite of the AMO Complete recall, the FDA says that many bottles of the contact lens solution are still on retail store shelves. Officials from the CDC say that millions of additional bottles may still be in the homes of contact lens wearers. Because of this, the FDA says that Advanced Medical Optics needs to do more to make consumers aware of the AMO Complete recall. The agency says that far less of the contact lens solution has been returned than it has expected, meaning that consumers may be unaware of the recall.
In spite of the AMO Complete recall, the FDA says that many bottles of the contact lens solution are still on retail store shelves. Officials from the CDC say that millions of additional bottles may still be in the homes of contact lens wearers. Because of this, the FDA says that Advanced Medical Optics needs to do more to make consumers aware of the AMO Complete recall. The agency says that far less of the contact lens solution has been returned than it has expected, meaning that consumers may be unaware of the recall.The FDA has asked Advanced Medical Optics to "redouble their efforts" in order to increase customer awareness of the AMO Complete recall. One means of doing this, the FDA says, would be to place advertisements about the contact lens solution recall in the same places where AMO Complete was advertised. But despite reports that consumers may still be using the contact lens solution, Advanced Medical Optics says that it currently has no plans to publish advertisements to alert consumers of the AMO Complete recall.
READ MORE LEGAL NEWS
Based on the CDC's investigation, Advanced Medical Optics issued a contact lens solution recall for AMO Complete. According to the agency, it has uncovered at least 138 confirmed cases of Acanthamoeba keratitis since beginning its investigation. One eye specialist estimated that as many as half of patients who develop the eye infection will eventually require a cornea transplant to correct the damage to their eyesight.
The Acanthamoeba keratitis infection is caused by organisms which are usually be found in water, soil or other places in the environment. According to estimates by the CDC, only one or two contact lens wearers per year develop the eye infection on average.
Written By Tony G
