LAWSUITS NEWS & LEGAL INFORMATION
Stevens Johnson Syndrome & Toxic Epidermal Necrolysis (SJS/TEN) Lawsuits
Read our Stevens Johnson Syndrome (SJS) FAQ
By Jane Mundy
Stevens Johnson syndrome/toxic epidermal necrolysis (SJS/TEN) is a severe skin reaction usually triggered by certain medications. Once thought to be separate conditions, Stevens-Johnson syndrome and toxic epidermal necrolysis are now considered part of a continuum. SJS is the least severe and TEN is the most severe of the disease spectrum. Some victims have filed SJS/TEN lawsuits seeking compensation for their injuries.
FREE STEVENS JOHNSON SYNDROME (SJS) LAWSUIT EVALUATION
Send your Stevens Johnson Syndrome (SJS) claim to a lawyer who will review your claim at NO COST or obligation.
GET LEGAL HELP NOW
GET LEGAL HELP NOW
Stevens Johnson Syndrome and Toxic Epidermal Necrolysis
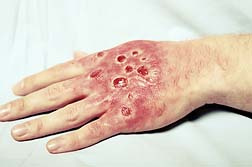 Stevens Johnson Syndrome (SJS), also known as Erythema Multiforme, Leyll's Syndrome, and in its later stages, Toxic Epidermal Necrolysis (TEN), is a serious and potentially life-threatening disease that causes large areas of the skin to become detached and lesions to develop in the mucous membranes.
Stevens Johnson Syndrome (SJS), also known as Erythema Multiforme, Leyll's Syndrome, and in its later stages, Toxic Epidermal Necrolysis (TEN), is a serious and potentially life-threatening disease that causes large areas of the skin to become detached and lesions to develop in the mucous membranes.Pharmaceutical manufacturers are aware of the risks associated with their drugs, yet proper warnings are for the most part inadequate. Many over-the-counter (OTC) drugs such as Ibuprofen—found in Advil and Motrin—have been linked to SJS. Without adequate warnings, patients who develop SJS may continue to take the SJS-causing drug, which could lead to TEN.
SJS/TEN symptoms usually begin 4-28 days after the onset of drug intake. Symptoms typically start with a fever and flu-like symptoms followed by a rash—usually starting on the face and eyes in particular, moving to the genitals—that can lead to blistering, severe peeling and open sores. Often an SJS rash is not diagnosed in time, and the condition becomes life-threatening. Other Stevens Johnson Syndrome symptoms include infections caused by the loss of skin or scarring of major organ systems that can be life-threatening.
Serious complications can include pneumonia, overwhelming bacterial infections (sepsis), shock, multiple organ failure, and death, according to the Centers for Disease Control and Prevention (CDC). About 10 percent of people with Stevens-Johnson syndrome die from the disease, while the condition is fatal in up to 50 percent of those with toxic epidermal necrolysis. A study published in the American Journal of Dermatology (Dec. 2015) found that the mortality rates for SJS and TEN are as high as 30 percent.
Although SJS/TEN is recognized as one of the few dermatological diseases that constitute a true medical emergency, as of 2015 no internationally accepted management guidelines had been established.
Treatment includes identification and withdrawal of the culprit drug, transfer to a specialist unit, supportive care, medical treatment, communication, and provision of appropriate information and emotional support.
SJS Medications
- Anti-gout medications, such as allopurinol
- Medications to treat seizures and mental illness (anticonvulsants and antipsychotics), with added risk if you also undergo radiation therapy
- Pain relievers, such as acetaminophen (Tylenol, others), ibuprofen (Advil, Motrin IB, others) and naproxen sodium (Aleve)
- Medications to fight infection, such as penicillin
The leading causes of this disease are the following medications:
- Ibuprofen (Advil, Children's Advil, Motrin, Children's Motrin and Nuprin)
- Bactrim , a sulfa drug
- Ketek , an antibiotic
- Dilantin , an anticonvulsant
- Zithromax , an antibiotic
- COX-2 inhibitors Bextra (Valdecoxib),Vioxx (Rofecoxib), Celebrex (Celecoxib), and Non Steroidal Anti-Inflammatory Drugs (NSAID's) including Daypro (Oxaprozein)
- Non Steroidal Anti Inflammatory drugs such as Feldene (Piroxicam), Naproxen (Aleve)
- Levaquin (levofloxacin), belonging to a class of drugs called fluoroquinolones, the antibiotic is used to treat bacterial infections such as pneumonia, bronchitis, sinus, skin, and urinary tract infections.
In January 2017, the US Department of Health and Human Services, the FDA and the Center for Drug Evaluation and Research (CDER) recommended that users of over-the-counter (OTC) acetaminophen-containing drugs should be warned in labeling that acetaminophen "may cause severe skin reactions" such as skin reddening, blisters and rash.
Several prescription and OTC medications have a warning label mandated by the FDA as a result of increased risk of developing Steven's Johnson Syndrome. The following drugs have either a new warning or additional wording on the label:
- Acetaminophen, (See FDA warning updated 2015)
- Tolectin 600 Tablets (Tometin Sodium)
- Tolectin DS Capsules (Tometin Sodium)
- Advil Allergy Sinus Tablets (200 mg ibuprofen / 30 mg pseudoephedrine HCI / 2 mg chlorpheniramine maleate)
- Advil Cold & Sinus Tablets (200 mg ibuprofen / 30 mg pseudoephedrine HCI)
- Advil Liqui-Gels (200 mg ibuprofen capsules)
- Advil Migraine Capsules (200 mg ibuprofen)
- Children's Motrin Chewable Tablets (50 mg ibuprofen)
- Motrin Junior Strength Chewable Tablets (100 mg ibuprofen)
- Motrin Cold & Sinus Tablets (200 mg ibuprofen / 30 mg pseudoephedrine HCI)
- Motrin IB Tablets (200 mg ibuprofen)
- Motrin Infants' Drops (50 mg / 1.25 mL ibuprofen oral suspension)
- Carbamazepine (marketed as Carbatrol, Equetro, Tegretol, and generics) particularly affecting Asian and South Asian patients
- Zithromax (see below)
Stevens Johnson Syndrome Symptoms
 Stevens Johnson Syndrome symptoms can begin with a fever, sore throat and headache and over a few days (or sometimes even minutes) can turn into skin lesions and blisters and respiratory infection. It generally affects the mucous membranes of the mouth and throat and swelling of the eyelids, and can progress to blindness and internal organ damage.
Stevens Johnson Syndrome symptoms can begin with a fever, sore throat and headache and over a few days (or sometimes even minutes) can turn into skin lesions and blisters and respiratory infection. It generally affects the mucous membranes of the mouth and throat and swelling of the eyelids, and can progress to blindness and internal organ damage.
The patient's skin burns from the inside out and often requires treatment in an intensive care unit (ICU) or the burn unit of a hospital. As Stevens Johnson Syndrome evolves, the skin sloughs off in sheets. If the skin lesions become infected, or the patient develops lesions in the lungs, it can result in death.
Recognizing early symptoms of SJS and providing prompt medical attention are the most valuable tools to minimize long-term damage.
In 2016 doctors thought a young boy had chickenpox after he was given Zithromax, a powerful antibiotic, for croup. Mail One (3/16/16) reported that the boy of three was left to fight for his life on a ventilator for nine weeks in the hospital.
Donna Rushing from Tampa told the Outbreak News This Week Radio Show (03/17/17) that her SJS symptoms started with “a flat, red rash on my face and my upper body” and recovery is not over. Her sense of taste has been affected, she has scars in her mouth and she also lost 60 pounds as a result of Stevens Johnson Syndrome.
Who Is at Risk for Stevens Johnson Syndrome?
Allergic drug reactions cause more than 100,000 deaths among patients in the United States each year, and adverse drug reactions are one of the leading causes of death in the US.
Adverse drug events (ADEs) account for more than 3.5 million physician office visits and 1 million emergency department visits each year, and preventable medication errors impact more than 7 million patients, according to the CDC (Sept 2016).
Those patients most likely to develop Stevens Johnson Syndrome are extremely allergic to antibiotics or painkillers like Ibuprofen. Most cases occur in adults between the ages of 20-40 years, but SJS has been diagnosed in children as young as three months old. Anyone who takes prescription or over-the-counter (OTC) drugs runs the risk of developing allergic reactions and developing serious health problems like Stevens Johnson Syndrome.
Stevens Johnson Syndrome Lawsuits
Lawsuits have been filed against a number of drug makers whose medications are allegedly linked to an increased risk of Stevens Johnson syndrome. One such lawsuit resulted in a $63 million award to the family of Samantha Reckis, who developed Stevens Johnson syndrome and toxic epidermal necrolysis when she was seven years old, after being given repeated doses of Children's Motrin. Reckis' family filed a lawsuit against Johnson & Johnson for failure to warn, and a jury awarded Reckis and her family $63 million. Johnson & Johnson appealed the award but the US Supreme Court refused to hear Johnson & Johnson's appeal.
Khaliah Shaw, age 26, was prescribed lamotrigine, an anticonvulsant drug sometimes prescribed for depression. After two weeks of taking the required dosage, Shaw was in "excruciating pain, she told local news station 11Alive. Shaw believes she was prescribed the wrong dosage of lamotrigine, which led to the development of SJS. She filed a lawsuit in early 2017, alleging her condition is the result of "pharmacists being too rushed, too busy, filling too many prescriptions," which can ultimately put the patient at risk. Shaw is looking for compensation to help with her $3.45 million medical bills for previous and future care related to SJS.
In 2016 defendant Johnson & Johnson prevailed in court after fighting a claim involving a young man who took his own life after taking Motrin and developing Stevens Johnson syndrome.
Also in 2016, the US Supreme Court upheld a huge award for an SJS plaintiff. Johnson & Johnson's application to appeal was rejected and a $63 million award handed to Samantha Reckis and her family after the young girl developed TEN, which was allegedly caused by her use of Children's Motrin.
A lawsuit was filed by Charlene Monk against Johnson & Johnson and Wal-Mart after her six-year-old daughter was diagnosed with SJS following a severe allergic reaction to medication. According to Courthouse News Service (11/4/16), Monk's daughter was treated with PediaCare Multi-Symptom Cold medication that contained acetaminophen in October 2014 and a few weeks later was given a generic children's ibuprofen to treat a fever. The case is Monk et al v. Johnson & Johnson et al, case number 1:16-cv-10273.
Register your Stevens Johnson Syndrome Case
If you or a loved one has suffered from Stevens Johnson Syndrome, you may qualify for damages or remedies that may be awarded in a Stevens Johnson Syndrome class action or lawsuit. Please click the link below to submit your Stevens Johnson Syndrome complaint to an attorney who will review your claim at no cost.Last updated on
STEVENS JOHNSON SYNDROME (SJS) LAWSUITS
- Zithromax SJS allegedly causing life threatening SJS, Stevens Johnson Syndrome skin inflammations.
- Levofloxacin SJS/TENS Resulting in Injury or Death
- Ketek allegedly causing potentially life threatening Stevens Johnson Syndrome (SJS) skin reaction.
- Dilantin SJS alleging that Dilantin causes this deadly skin inflammation.
- Bactrim allegedly causing SJS, Stevens Johnson Syndrome skin inflammations that can be life threatening.
STEVENS JOHNSON SYNDROME (SJS) LEGAL ARTICLES AND INTERVIEWS
Woman's Skin Melts Off From Stevens-Johnson Syndrome
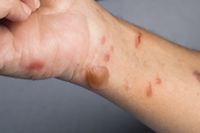
Stevens Johnson Syndrome Victim Has Permanent Scarring in her Mouth
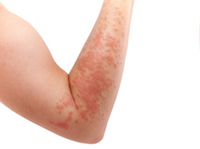
Texas Woman Suffers Stevens Johnson Syndrome from Bipolar Medication


May 13, 2017
Snellville, GA: A Georgia woman claims that her skin melted off when she contracted Stevens-Johnson Syndrome (SJS) after being given an incorrect dosage of an antidepressant by a pharmacist. READ MORE
Stevens Johnson Syndrome Victim Has Permanent Scarring in her Mouth

April 17, 2017
Tampa, FL: Donna Rushing is a resident from Tampa who developed Stevens Johnson Syndrome from a medication she had been taking. While she was spared hospitalization and more serious SJS side effects that can often include massive loss of skin and life-threatening infections, her battle with Stevens Johnson skin disease was enough to cause her to lose weight, affected her eating and has left her with permanent scarring in her mouth. READ MORE
Texas Woman Suffers Stevens Johnson Syndrome from Bipolar Medication

March 19, 2017
Houston, TX: The majority of patients and plaintiffs touched by the scourge of Stevens Johnson Syndrome (SJS) can usually track, or at the very least suspect with a reasonable hypothesis that Stevens Johnson Syndrome symptoms materialize through the use of pharmaceutical drugs and medications including standard, everyday products such as over-the-counter (OTC) pain medication. READ MORE
READ MORE Drugs/Medical Settlements and Legal News
READ MORE Personal Injury Settlements and Legal News
READ MORE Personal Injury Settlements and Legal News

READER COMMENTS
Heather Caldwell
on
HC, Chicagoland
on
Fast forward to opioid trials, topamax, gabapentin, etc. And resultant migraines and double vision. Then epidurals and even multiple nerve ablations to dull the pain. Finally get my meds, therapy, army of docs lined up, and the government starts taking away and/or lowering the dosage of the only two meds that allowed me to live a halfway normal life.
I have consulted a well known allergist. I asked him if I could try some of the drugs to see if the sensitivity had worn off after several years. He adamantly told me I COULD DIE if I were to do that.
Where my rheumatologist at the time had me schlepping an epi pen with me, the allergist told me to toss it. With the type of reactions I'd had to these drugs, no epi pen was going to help.
I am now in more pain than ever, trying to advocate for myself and be a full time caregiver to a loved one who has been through a terrible physical ordeal and requires a LOT of physical help as he recovers. ...and whose pain meds have also been screwed with, which makes things more difficult for both of us.
Side note: a few yrs. ago one of my doctors really wanted me to try one of the anti depressant drugs that's also used for pain control. When I called Eli Lilly they would not disclose the frequency of SJS or TENs cases associated with their drug. I had to employ the FREEDOM OF INFORMATION ACT to get the data. The next year I started noting references to SJS symptoms in their warning copy. I now need to take a pill for high blood pressure. Available research tells me the SJS rate is higher than I'm comfortable with. Here we go again, but at least the discussion this time will be with an internist.
PS: Like one of the other contributors, I also wound up with neuropathy, cause unknown, after becoming an SJS'er.
Also, I was told by a law firm in 2005 that basically because I hadn't died or been injured enough, I wouldn't be able to join any of the large lawsuits suing the manufacturer of Bextra.
Melissa D Reid
on
Anne
on
Lovella
on
Jennifer
on
Jerry Lowe
on
I was sent to a burn center and was told that I had SJS and they removed my remaining skin. 55% damage to skin and liver damage, plus 20% loss of cornea in the left eye.
I am finding it difficult to find legal representation in Ohio where I live in order to seek compensation, due to the fact that I am not able to work. If anyone has legal advice please email me so I can get support.
Nancy
on
Ron Cobb
on
Mary Ann Town
on
Ricardo Vasquez
on
Ricardo Vasquez
Love kept me alive
God kept me alive
Good Doctors keeping me alive
Randolph R Perry Sr
on
Debbie
on
Missouri
on
Oklahoma
on
California
on
Oklahoma
on
Texas
on
New York
on
Idaho
on
Texas
on
California
on
Illinois
on
Kentucky
on
California
on
Respiratory problem, skin disorder, blurred vision. Were told feet may have to be amputated, on breathing machine to a tube in his throat. Lost 45lbs.
Alabama
on
Alabama
on
Stephen johnson syndrome.
Ontario
on
Florida
on
South Carolina
on
North Carolina
on
Ontario
on
Unable to describe well. Would like to speak with someone pls.
North Carolina
on
Arizona
on
Pain, out of work for a week.
Florida
on
Texas
on
I have permanent skin discolorization, scarring on my arms and legs. I have massive blisters on the bottoms of my feet preventing me from being able to walk for long period of times.
New York
on
Washington
on
Tennessee
on
Washington
on
Virginia
on
California
on