LAWSUITS NEWS & LEGAL INFORMATION
Defective Knee Implants
Were you looking for 3M Bair Hugger Infections lawsuits?
By Jane Mundy
Allegedly defective knee implants have been a cause of concern for medical professionals and patients alike, with some knee implant devices being the subject of recalls due to a high rate of complications, ranging from infection and trauma to loosening or poor implant positioning, along with a defective medical device. Among issues allegedly associated with knee implant failure are debonding of the joints, pain in and around the knee, decreased mobility and range and other problems often leading to revision surgery. Many defective knee implant lawsuits involve metal-on-metal devices.
FREE DEFECTIVE KNEE IMPLANT LAWSUIT EVALUATION
Send your Defective Knee Implant claim to a lawyer who will review your claim at NO COST or obligation.
GET LEGAL HELP NOW
GET LEGAL HELP NOW
 About 1.2 million hip and knee surgeries are performed in the US each year and as baby boomers age, up to 4 million implants per year are predicted. Most knee implants are supposed to last from 15-20 years but the failure rate is estimated at 10 percent and often because the knee implant is defective, according to the American Association of Orthopedic Surgeons. Eight percent of knee replacements lead to costly and painful revision surgery.
About 1.2 million hip and knee surgeries are performed in the US each year and as baby boomers age, up to 4 million implants per year are predicted. Most knee implants are supposed to last from 15-20 years but the failure rate is estimated at 10 percent and often because the knee implant is defective, according to the American Association of Orthopedic Surgeons. Eight percent of knee replacements lead to costly and painful revision surgery.Consumers Union urged medical device manufacturers, including Biomet, Inc., DePuy Synthes, Smith & Nephew, Stryker Corporation, Wright Medical Technology, Inc, and Zimmer Holdings to provide a warranty to include the following:
- Covers the full cost of replacing the flawed device, including the device, surgeon and hospital costs as well as related patient out-of-pocket costs.
- Establishes a clear system for patients to use, including a toll-free phone line and a registration number to track the claims process, with physicians charging the device company, not the patient.
- Does not eliminate the patient' right to sue if they use a warranty.
- Provides the patient with a full explanation if a warranty claim is denied and an appeals process for denials.
- Does not disqualify patients across the board if they have specific diseases or illnesses that are not related to the failure of the device.
Zimmer NexGen
Issues with the Zimmer NexGen CR-Flex Porous Femoral component allegedly include loosening of the implant and failure of the knee implant, which could require revision surgery for repair.
Symptoms of loosening of the knee implant include pain and limited mobility in the affected joint. Patients who believe they are having problems with their knee replacement are advised to have a bone scan (rather than an x-ray, which will not show loosening of the knee implant).
Lawsuits have been filed against Zimmer regarding various NexGen Knee implants alleging the NexGen high-flex knee implants are defective and associated with unnecessary injury to patients. As of September, 2013 there are 1,035 cases pending in the Zimmer NexGen knee implant Multidistrict Litigation (MDL 2272, US District Court for the Northern District of Illinois).
In August 2013 a lawsuit filed in Texas Eastern District Court (case no. 5:2013cv00095) claims the Zimmer NexGen knee implant is defective due to loosening. The plaintiff had to undergo revision surgery. A Nevada resident suffered the same complications, underwent revision surgery and filed a defective knee lawsuit (case no. 2:2013cv01975, Nevada District Court).
Stryker ShapeMatch Cutting Guide
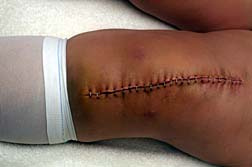 The Stryker ShapeMatch Cutting Guide is used during total knee replacement surgery to put the replacement components in place and guide the surgeon when marking the bone before the bone is cut. The Stryker ShapeMatch Cutting Guides are single-use devices used with the Triathlon Knee System. They were designed to allow for smaller incisions in the knee and to customize the surgery for each patient's anatomy.
The Stryker ShapeMatch Cutting Guide is used during total knee replacement surgery to put the replacement components in place and guide the surgeon when marking the bone before the bone is cut. The Stryker ShapeMatch Cutting Guides are single-use devices used with the Triathlon Knee System. They were designed to allow for smaller incisions in the knee and to customize the surgery for each patient's anatomy.The Stryker cutting guides involve the use of an MRI prior to surgery so that a three-dimensional model of the knee can be made. The 3D model is used to determine the size and position of the knee implant. Once those are determined, the ShapeMatch technology is used to create a customized guide to aid the surgeon in making incisions and implanting the knee.
There are concerns, however, that the device can be misaligned, causing knee implants to be placed in the wrong position, resulting in joint instability, pain, limited mobility and fractures. The US Food and Drug Administration (FDA) noted when it announced a Class I Urgent Medical Device Recall of the Stryker ShapeMatch that it had received 44 reports of incidents linked to the ShapeMatch.
Zimmer Natural Knee Implant
Smith & Nephew Knee Implants
In March, 2010 the FDA recalled Journey II Uni Tibial Baseplates after Smith & Nephew received complaints about baseplates breaking, which requires revision surgery. The Class II recall affected approximately 40,000 Journey II knee implants.
In January 2014 the Australian Government' Department of Health issued a safety alert, advising consumers and health professionals that Smith & Nephew has issued a hazard alert regarding the femoral implant component of the Journey Bi-Cruciate Stabilised (BCS) knee replacement system. This particular knee replacement system has been recalled by the Australian Therapeutic Goods Administration (TGA).
DePuy Knee Implants
Symptoms of Defective Knee Implant
Defective Knee Implant Lawsuits
Defective Knee Implant Legal Help
If you or a loved one has suffered damages or injuries from a Defective Knee Implant, please click the link below and your complaint will be sent to a Medical Device lawyer who may evaluate your claim at no cost or obligation.
Last updated on
DEFECTIVE KNEE IMPLANT LAWSUITS
- Exactech Optetrak Knee Replacement Failure Lawsuit
- Smith & Nephew Defective Knee Replacements
- DePuy Synthes Attune Knee Replacement Lawsuit lawyers investigating complaints involving premature failure
- Zimmer Class II recall of the Zimmer Persona Trabecular Metal Tibial Plate
- Stryker Triathlon Knee Implant Injury using Shapematch Cutting Guide
- Zimmer Natural alleging defective Zimmer Natural knee and Natural hip replacements.
- Zimmer NexGen alleging defective knee replacements.
- Hip & Knee Replacement alleging sterilization practices can cause implant failure.
- Knee Replacement alleging defects in certain Oxinium knee replacements.
DEFECTIVE KNEE IMPLANT LEGAL ARTICLES AND INTERVIEWS
Tentative Settlement Reached in Zimmer NexGen Knee-Implant MDL
DePuy Faces Yet Another Lawsuit Concerning its Attune Knee Replacement System

Tracking Systems in the U.K Key to Care for Hip Implant Patients, But Non-Existent in the U.S.


February 15, 2018
Los Angeles, CA: Zimmer, the maker of NexGen knee replacements and the defendant in a defective products multi-district litigation (MDL) has reached a preliminary settlement with the plaintiffs in the longstanding MDL. READ MORE
DePuy Faces Yet Another Lawsuit Concerning its Attune Knee Replacement System

January 28, 2018
Jackson, MI: Whenever an individual files a defective knee implant lawsuit, the focus is normally – and correctly – on the failed component due to an alleged design, or manufacturing defect. But a companion issue just as devastating as the failure of the implant is, for the patient, a requirement for revision surgery to eradicate the problematic implant and replace it with another one. Revision surgery is usually, a more complex procedure. READ MORE
Tracking Systems in the U.K Key to Care for Hip Implant Patients, But Non-Existent in the U.S.

January 26, 2018
Santa Cruz, CA:The study, published in the Bone & Joint Journal, finds that the rate of metal-on-metal (MoM) hip implant patients undergoing revision has increased over time. Researchers at the Universities of Oxford and Bristol have suggested the observed differences are in part due to increased patient surveillance. In the U.S., patient surveillance is pretty much left up to the patient. READ MORE
READ MORE Defective Products Settlements and Legal News
READ MORE Drugs/Medical Settlements and Legal News
READ MORE Personal Injury Settlements and Legal News
READ MORE Drugs/Medical Settlements and Legal News
READ MORE Personal Injury Settlements and Legal News

READER COMMENTS
debra monsoor
on
Janyce Westerman
on
Kristine Thatch
on
Lisa Ramsey
on
Louis Huther jr
on
Ralph
on
Scott
on
Never stopped hurting , swelling , barley walking.. bastard orthopedic admitted nothing , won't even admit it's a problem .radiologist. said knee looks like extreme trauma , shave you been in car crash ? No I have not ,just bad hardware ,and horrible doctor
Irene Carnahan
on
Rosita wheeler
on
GWEN WILKERSON
on
Cericola
on
JOYCE
on
Christina Smith
on
TLM
on
My surgeon was confident about the Zimmer Persona (cemented version), that would work great for me. Immediately I noticed issues and had extreme pain, and was told that was normal. The pain never got better, my range of motion was not good, I even went thru an MUA three months after the original surgery, but afterwards my knee froze up again, and was very painful to get it past 100 degrees.
At the 3-month post op appointment, my surgeon wasn't concerned about my pain or range of motion. He stated that it may be as good as it was going to get. I didn't see him again until my one year follow-up appointment.
I still had to walk with a cane, and could only put weight on my knee for a few minutes before the pain was unbearable. He It was verified that tibial component was loose. In just over a year I went thru a revision.
I requested my medical records & surgical notes, it stated that the bone cement (no brand) did not adhere to the bone. I had to call the office to find out the brand of bone cement, which was Zimmer.
I have searched for information on bone cement recalls, and contacted several online medical sites trying to find out if there is a recall, but I have not been able to find out anything on Zimmer bone cement, and if there has been any recalls.
Barbara grzanecki
on
Janie Gaston
on
Neale Humes
on
alma sliger-clark
on
April Moses
on
Tina Johnson
on
alma clark
on
I hope you don't ignore me the brand of my replacement was EXACTECH