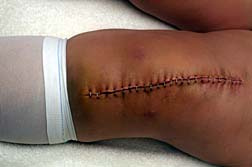
 The FDA is now concerned enough that it has sent warning letters to the president of Stryker Biotech, warning that the company's practices, "…are not in conformity with the Current Good Manufacturing Practice requirements of the Quality System regulation." At least three warning letters have been sent to the company regarding the issue. The FDA says that deficiencies at Stryker's New Jersey manufacturing facility resulted in the manufacture of defective hip replacement components, causing injury to patients who received the defective components. Some of the patients who received defective parts required further surgery to replace those parts.
The FDA is now concerned enough that it has sent warning letters to the president of Stryker Biotech, warning that the company's practices, "…are not in conformity with the Current Good Manufacturing Practice requirements of the Quality System regulation." At least three warning letters have been sent to the company regarding the issue. The FDA says that deficiencies at Stryker's New Jersey manufacturing facility resulted in the manufacture of defective hip replacement components, causing injury to patients who received the defective components. Some of the patients who received defective parts required further surgery to replace those parts.According to one letter from the FDA, sent on November 28, 2007, Stryker received "continual complaints" starting in January 2005, regarding hip implant components that had poor fixation, made squeaking noises and were improperly seated, resulting in bone fractures. Further complaints included patients experiencing pieces of implant parts breaking off, difficulty walking and pain. The FDA goes on to note that Stryker has not yet taken adequate action to correct the problems.
During a six-week inspection of Stryker's New Jersey plant, the FDA found numerous issues, including instances of Staphylococcus bacteria, the pathogen that causes staph infections. The FDA's letter notes that Stryker, "has not identified the root causes of the microorganism contamination and has not executed corrective and preventive action to prevent recurrence."
What this means for patients who received hip implants by Stryker is that they may have received faulty parts. It may also mean that they require additional surgeries to correct any implant defects they have been exposed to.
READ MORE HIP AND KNEE REPLACEMENT LEGAL NEWS
Along with the addition surgeries come additional medical bills, additional time off work, and additional recovery time, not to mention the additional time spent in pain and struggling to walk. All that can add up to one big headache for patients who were given defective hip or knee implants.
If you were implanted with defective hip or knee parts you may be eligible to join a lawsuit against the manufacturer of the defective parts. Contacting a lawyer to discuss your options would be a good idea.

READER COMMENTS
Eugene Sharp
on
Eugene Sharp
on
Stryker Triathlon Knee Implant is a horrible device it does not work, it is Dangerous to people and needs to be recalled by the FDA IMMEDIATELY!!!!!!!
I filed a adverse event complaint with the FDA yesterday on this Stryker Triathlon Knee Implant. FDA said if they get 2 to 3 or 4 complaints or more then they will really stand up and take notice.
everyone who has this problem with the STRYKER TRIATHLON KNEE IMPLANT NEEDS TO FILE AN ADVERSE EVENT REPORT WITH THE FDA ON THIS PRODUCT SO WE CAN GET THIS PRODUCT RECALLED AND OFF THE MARKET!!!!! THEN WE CAN HOLD STRYKER ORTHOPEDICS RESPONSIBLE FOR ALL OF THE DAMAGE THEY HAVE DONE TO ALL OF US!!!!
Larry B. Dorsey
on
Sandy
on
Jerry Zigmund
on
Beverley
on
Phyllis
on
B.T.
on
S ws
on
TWB
on
J B
on
J G
on