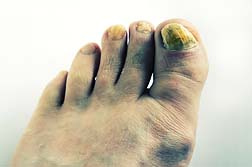
 Lamisil has been around since 1996, and is available in oral tablet, topical cream, gel, and solution—all given FDA approval across various years starting in 1996. The latest sibling to come onto the market has been Lamisil oral granules, which were given FDA approval on September 28th of last year and is available in 125 mg, and 187.5 mg versions.
Lamisil has been around since 1996, and is available in oral tablet, topical cream, gel, and solution—all given FDA approval across various years starting in 1996. The latest sibling to come onto the market has been Lamisil oral granules, which were given FDA approval on September 28th of last year and is available in 125 mg, and 187.5 mg versions.The type of fungus experienced, will usually dictate the version of Lamisil that is prescribed. No one likes to live with fungus, which is not pleasant. And Lamisil has proven effective in getting rid of it. The problem appears to be the adverse effects, and the seriousness of them.
For example, since the various forms of Lamisil have appeared through to early January of this year, there have been a total of 4,079 reports of adverse reaction of one form or another in the US. Of those 2,876 have been described as 'serious' enough to involve death, disability, initial or prolonged hospitalization, life-threatening events, congenital anomaly or other serious Lamisil side effects. That's more than half, considered serious, for all persons across all demographics.
In children under 16 years of age, the 'serious scale' is more alarming. Of 48 adverse reaction reports in the US, 45 are considered 'serious.' That's a huge percentage. And while there have been no deaths reported in the US, concern is mounting.
And that concern has to do with adverse effects that aren't identified on product labeling—specifically, psychiatric reactions. The most common Lamisil side effect continues to be skin reaction, and while some of those skin reactions are indeed, in of themselves serious (including Stevens-Johnson syndrome), what has the FDA sitting up and taking notice are the reports of psychiatric events.
An FDA staff report was prepared in anticipation of a meeting of the FDA Pediatric Advisory Committee scheduled for yesterday (November 18th), which was expected to recommend continued monitoring, and product labeling changes to include reference for potential psychiatric events.
While the staff report breaks out the various instances of skin rash and other skin reactions, neurological and psychiatric events are also listed in relation to the pediatric use of Lamisil. These include 3 cases of seizures and shaking, headache and neck pain, mental impairment, difficulty walking, and somnolence.
One 9-year-old girl found that she had difficulty doing math after taking Lamisil for five days. Once the medication was stopped, she fully recovered.
Additionally, there were 3 reports of psychiatric events, which ran the gamut from suicidal ideation, depression and self-harm. In one case the patient had also been diagnosed with Lyme Disease that carried cerebral involvement, possibly contributing to the symptoms. However, aside from the latter all three patients recovered when the Lamisil treatment was stopped.
READ MORE LAMISIL LEGAL NEWS
The remaining 41 cases were considered serious because the reporter considered the event to be medically significant.
Because the proportion of serious Lamisil side effects is far greater in children than it is in adults, the FDA is continuing to investigate. While some may argue that using a topical Lamisil cream is a less-direct pathway than oral tablets, the natural ability of the skin to absorb and transfer chemicals raises the concern that using Lamisil cream to treat Lamisil fungus takes the treatment beyond the initial fungus with, in some cases, serious consequences.
Lamisil is made by Novartis.

READER COMMENTS
Steve Scott
on
One should listen to independent biologists, chemists, and scientists, and former pharma lab workers that develop and test these drugs to really gain some insight on just how few drugs have positive long-term effect vs. the short and long-term risks(and better yet to explain why and why not, scientifically, and to demonstrate a huge lack of knowledge on the part of the general MD community, and the complicity of the AMA and the FDA to keep the health industry profitable.
We won't be going back to that doctor. He wouldn't listen even if we took him living(or dead) proof. He is a doctor, with a license and a med degree. Imagine if he had to face the idea that all of the time and money he spent to attain that status was mostly a facade.
Whether though ingnorance or not,on his part, I see it as attempted physical assault even if he is generally immune from criminal malfeasance. The FDA and the AMA will lose control of the profit generators only when people begin to stop groveling at the feet of doctors and challenge the logic and real long established science & biology, and hold them accountable. Not so much with law suits, but challenge with facts, and if they are not then willing to learn about a subject because they already know it all........then walk on the spot.
I personally would not pay either --incompetent(or lack of) service and knowledge, does not deserve rewards.
When it finally affects their business practice enough, they can go flip burgers for a living, and tell their high school shift manager exactly what they think they know...I am sure he will care.
Amy Shea
on
Brigid Hynes
on