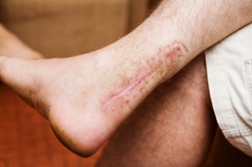
 According to the Madison County Record of 8/9/10, plaintiffs allege they didn't know about the risk for tendon rupture, especially among patients older than 60 years, or those on corticosteroid therapy.
According to the Madison County Record of 8/9/10, plaintiffs allege they didn't know about the risk for tendon rupture, especially among patients older than 60 years, or those on corticosteroid therapy.The lawsuit claims that when Levaquin first appeared on the market in 1997, references to the potential for tendon ruptures were buried in a substantial list of side effects. Plaintiffs also claim that warnings failed to advise that the risk for tendon injury tripled for those in the high-risk group.
In 2002, according to the complaint, the Levaquin label was updated to include warnings with regard to corticosteroid therapy but failed to mention the risks to the elderly.
"Accordingly, despite the 2002 label change, Levaquin prescriptions only increased and tendon injuries mounted," the suit states.
Johnson and Johnson Pharmaceutical Research and Development (PRD initiated a study on the effects of Levaquin in 2001. However it is alleged that the defendant limited its analysis to Achilles tendon ruptures and failed to assess the risk of tendon toxicity. It is also alleged, according to the text of the lawsuit, that Johnson and Johnson PRD produced an algorithm that excluded close to 70 percent of health claims for the elderly who suffered Achilles tendon rupture.
READ MORE LEVAQUIN LEGAL NEWS
As a result, the Johnson and Johnson PRD study found no increased risk of Achilles tendon rupture, and neither age nor corticosteroid use altered the findings.
There were 1,044 reports of tendon problems, with 282 reports of tendon ruptures from 1997 through 2005, according to the complaint. Named as defendants in the action are Johnson and Johnson, Ortho-McNeil Pharmaceutical, Johnson and Johnson Pharmaceutical Research and Development (PRD) and Walgreens.
