 Just one month prior to the recall, U.S. marshals seized heart valves and other implantable medical devices made from cow and pig tissue by the New Jersey company because of concerns about their sterility. The products include pediatric heart valves and conduits, or tube-like devices for blood flow, as well as surgical patches, arterial grafts and annuloplasty rings used to help repair heart valves. At that time, Shelhigh refused to perform a voluntary recall of its products, even though its own records indicated a number of sterility test failures and that its testing and retesting procedures were not properly performed.
Just one month prior to the recall, U.S. marshals seized heart valves and other implantable medical devices made from cow and pig tissue by the New Jersey company because of concerns about their sterility. The products include pediatric heart valves and conduits, or tube-like devices for blood flow, as well as surgical patches, arterial grafts and annuloplasty rings used to help repair heart valves. At that time, Shelhigh refused to perform a voluntary recall of its products, even though its own records indicated a number of sterility test failures and that its testing and retesting procedures were not properly performed.According to the [FDA] "the manufacture, packing, storage, and installation [of Shelhigh products] are not in conformity with current good manufacturing practice (CGMP) requirements."
The "Complaint for Forfeiture" issued by the FDA states that the medical devices made by Shelhigh are cardiovascular, neurological, and general surgery sterile implantable medical devices, including pulmonic and aortic heart valves and pericardial patches that are surgically implanted, which are used in adults and infants.
All the devices manufactured by Shelhigh must go through a No-React® rinse after sterilization. The purpose of this final rinse process is to stabilize the sterilant to prevent it from leaching off the device and causing an allergic reaction in patients. This No-React® process is performed in the firm's 'class 100 aseptic manufacturing room' (class 100 room), to prevent contamination of the sterile devices. But the FDA's inspection in October and December of 2006 revealed that the procedures were not adequate.
Furthermore, the agency found that Shelhigh "does not have a procedure describing how samples used to test for sterility are processed and controlled prior to sterilization to ensure that they are representative of finished devices".
Shelhigh also allowed at least four lots of devices that failed sterility testing to be released for distribution in the last two years.
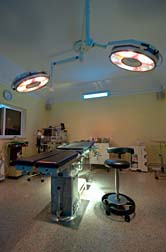
Sterile Environment?
This No-React® process is performed in the class 100 room. This is what was found in class 100 room:
- no separate gowning room for employees to dress in sterile garments before entering the room
- Employees do not wear sterile shoe covers before entering or when inside the class 100 room. In the class 100 room, the entire body should be covered by sterile garments. Sterile shoe covers are necessary to cover shoes, which are recognized vectors for transporting high levels of microorganisms throughout a facility;
- A non-sterile stainless steel cart is transported into the class 100 room during product processing
- Pieces of raw and painted wood were observed in the firm's class 100 and class 1,000 rooms. Wood is porous, difficult to disinfect, can allow for the growth of bacteria and mold and contamination of the environment.
- Ceiling panels stained with unknown substances;
- Dislodged ceiling panels; portions of ceiling panels missing;
- Air diffusers with corrosion and dirt accumulation.
The FDA is recommending physicians and hospital officials consider using alternative products.
"Since these are critical devices implanted into seriously-ill patients, ensuring their sterility is absolutely essential to prevent infection," said Dr. Daniel Schultz, director of the FDA's Center for Devices and Radiological Health.
The U.S. Food and Drug Administration (FDA) today issued a formal written request to Shelhigh, Inc. to recall all of its medical devices remaining in the marketplace, including hospital inventories, because of sterility concerns.
On April 17, 2007, U.S. Marshals, at FDA's request, seized all medical devices including components at Shelhigh's Union, N.J. facility after finding significant deficiencies in the company's manufacturing processes. During the seizure, Shelhigh was asked to perform a voluntary recall of its products, but the company declined.
FDA recommends that doctors and hospitals consider using alternative products. Physicians and patients concerned about Shelhigh devices can visit two of their web pages on this subject: fda.gov/cdrh/medicaldevicesafety/atp/041907-shelhigh.html and fda.gov/cdrh/safety/041907-shelhigh.html for more information, including a list of the company's products.
"Since these are critical devices implanted into seriously-ill patients, ensuring their sterility is absolutely essential to prevent infection," said Daniel Schultz, M.D., director, FDA's Center for Devices and Radiological Health. "FDA will continue to provide up-to-date information to patients and physicians about this ongoing public health matter."
The company's deficiencies, described in a complaint filed with the U.S. District Court of New Jersey, may compromise the safety and effectiveness of the devices. Shelhigh's own records indicate a number of sterility test failures and that its testing and retesting procedures were not properly performed.
Shelhigh devices are used in infants, children and adults. The products include pediatric heart valves, tube-like devices for blood flow (conduits), surgical patches, dural patches to aid in tissue recovery after neurosurgery, annuloplasty rings to help repair heart valves, and arterial grafts.
