Posts Tagged ‘ NECC ’
UPDATE 2: Multi-State Fungal Meningitis Outbreak by the Numbers
October 9th, 2012. By AbiK
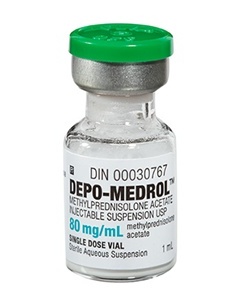 (Updated Information as of 10/12/12 shown in RED below)
(Updated Information as of 10/12/12 shown in RED below)
Here’s the latest on the multi-state fungal meningitis outbreak.
– Reuters is reporting a member of the U.S. Senate has requested the U.S. Justice Department to conduct a criminal probe of possible fraud violations by NECC, the supplier of the contaminated steroid injections.
-Also, new estimates suggest as many as 14,000 individuals may have received the contaminated steroid—previously that number had been estimated to be 13,000.
-See updated ‘by the numbers’ information below.
If you or someone you know have had a steroid injection—an epidural (according to the CDC, epidurals given for childbirth do not use the same medication involved in this outbreak), a shot given by an orthopedist, etc.—check with your doctor and/or healthcare facility to find out if you may have been affected. Also, be aware of the symptoms associated with meningitis (some are listed below) and seek immediate medical help should you suspect that you may have been affected by a contaminated steroid injection.
While the source of the contamination that has led to so many cases of fungal meningitis has not yet been determined, an investigation continues. Meningitis victims may also wish to seek legal help and can do so by submitting the form here.
Most importantly though, be sure to talk to your medical provider if you suspect you may have received a tainted steroid injection. Symptoms of meningitis include new or worsening headache, fever, sensitivity to light, stiff neck, new weakness or numbness in any part of your body, slurred speech, and/or increased pain, redness or swelling at the injec tion site.
tion site.
Here, the latest numbers on the meningitis outbreak:
Date of Recall:
September 26, 2012
Number of Lots Recalled:
3 lots of preservative-free methylprednisolone acetate (80mg/ml); the 3 lots contained 17,676 vials of medicine (Reuters)
Number of Suppliers who have Issued a Recall:
1—New England Compounding Center (NECC) issued a voluntary recall on September 26, 2012
Start Date in which Recalled Lots of Steroid Injections may have been used:
May 21, 2012
Number of States with Healthcare Facilities that received Recalled Lots of Steroid Injections:
23 (California, Connecticut, Florida, Georgia, Idaho, Illinois, Indiana, Maryland, Michigan, Minnesota, North Carolina, New Hampshire, New Jersey, Nevada, New York, Ohio, Pennsylvania, Rhode Island, South Carolina, Tennessee, Texas, Virginia, West Virginia)
First State to Detect a Case of Fungal Meningitis related to the Recalled Steroid Injections:
Tennessee, which has had the most reported cases to date (35) and 4 reported deaths
Number of States with Reported Cases of Fungal Meningitis:
11 states have reported cases–Florida (4), Idaho (1), Indiana (12), Maryland (8), Michigan (25), Minnesota (3), North Carolina (2), Ohio (1), Tennessee (39), Virginia (24), New Jersey (1)
Number of Reported Cases of Fungal Meningitis:
170 cases of fungal meningitis have been reported to date
Number of Deaths related to Fungal Meningitis Outbreak:
14 deaths have been reported to date in the following states: Florida (2), Indiana (1), Maryland (1), Michigan (3), Tennessee (6), Virginia (1)
Number of Individuals who may have Received an Injection from the Recalled Lots:
An estimated 14,000 individuals may have received a steroid injection from the recalled lots
Source: Centers for Disease Control (CDC.com), unless otherwise noted. A full list of healthcare facilities by state that had received part of the recalled lots from NECC is available at the CDC website.
Archive by Category
- Accidents (24)
- Airlines (9)
- Asbestos Mesothelioma (262)
- Automotive (25)
- Celebrity (14)
- Class Action (84)
- Complaints/Comments (15)
- Consumer Fraud (84)
- Contest (2)
- Court of Public Opinion (5)
- Crazy Sh*t Lawyers See (61)
- Criminal Law (4)
- Defective Products (111)
- DePuy ASR Hip Recall (2)
- Discrimination (22)
- Drugs/Medical (248)
- Elder Care Abuse (4)
- Emerging Issues (462)
- Employment (54)
- Environment (52)
- Financial (28)
- Food Illness (15)
- Human/Civil Rights (4)
- Insecurities (5)
- Insurance (16)
- Intellectual Property (16)
- Internet/E-commerce (19)
- lawsuits (161)
- Lawyers (20)
- Lawyers Giving Back (43)
- Lex Levity (10)
- Personal Injury (106)
- Pleading Ignorance (53)
- Real Estate (2)
- Recall (6)
- Scam (3)
- Securities (13)
- Settlement (81)
- Tort Reform (2)
- Totally Tortelicious (81)
- Veterans (11)
- Whistleblower (9)