Posts Tagged ‘ DES Daughters ’
Concerned about DES Exposure? Take this DES Self-Assessment
August 20th, 2012. By AbiK
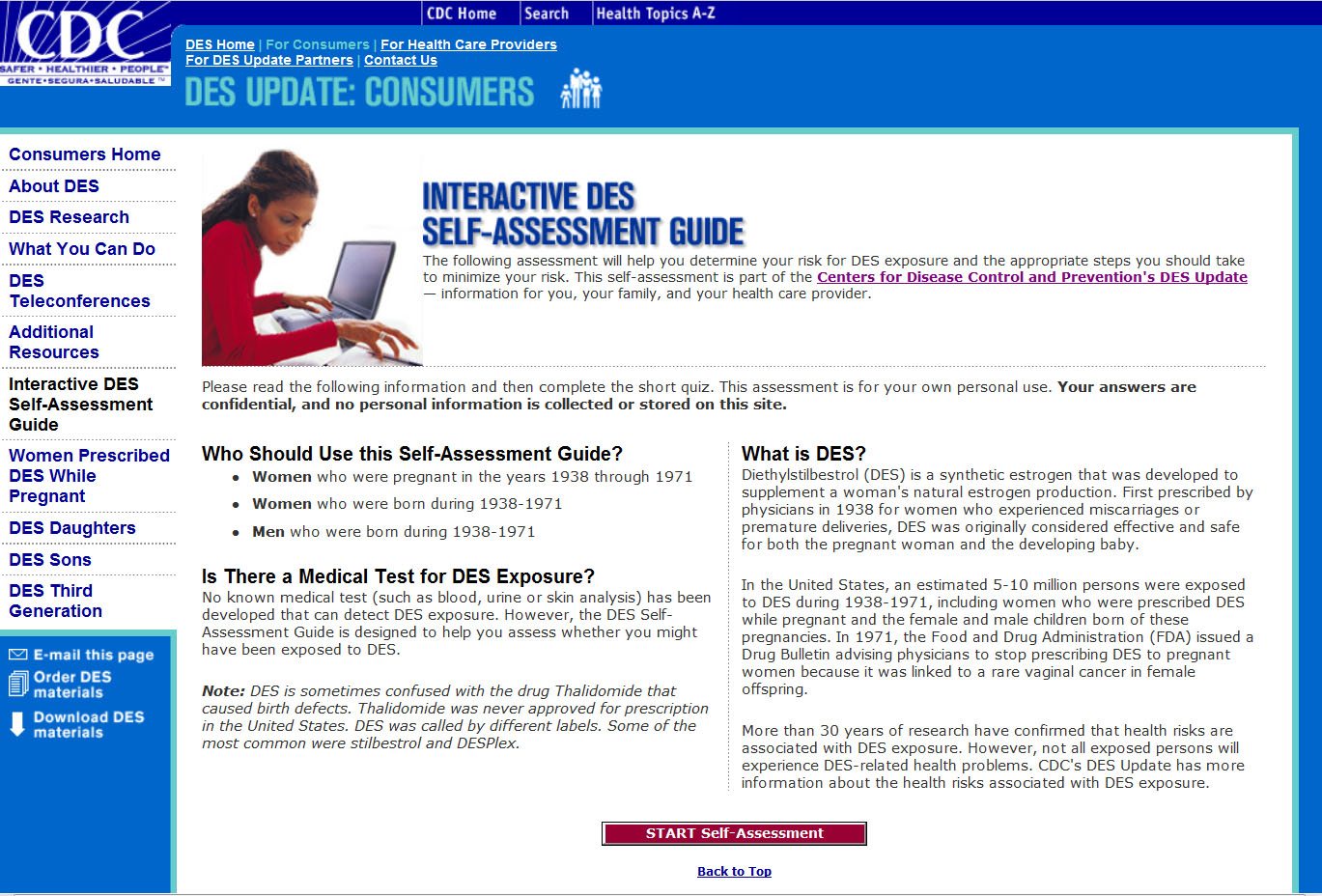 Pregnancy drug DES has been back in the media spotlight lately as more and more women become aware of possible DES exposure they may have experienced in utero. DES—or diethylstilbestrol—was prescribed to pregnant women during between the 1940s and the early 1970s to help prevent miscarriage.
Pregnancy drug DES has been back in the media spotlight lately as more and more women become aware of possible DES exposure they may have experienced in utero. DES—or diethylstilbestrol—was prescribed to pregnant women during between the 1940s and the early 1970s to help prevent miscarriage.
DES was ultimately removed from the market when studies revealed the drug’s link to certain cancers, among other potential side effects. DES risks include vaginal and cervical cancers for women whose mothers had taken diethylstilbestrol during pregnancy. Additionally, harmful DES side effects may include breast cancer, early menopause and infertility in women who had actually taken the drug.
The Centers for Disease Control and Prevention (CDC) has a DES Consumer Guide that provides information about DES. The web page includes a DES Self-Assessment Guide that helps indicate whether an individual has had the potential to have been exposed to DES and the possible DES health risks if, indeed, DES exposure did occur. Please note: the DES Self-Assessment Guide cannot specifically determine whether a person was actually exposed to DES, but it does provide a guide for further discussion with a healthcare professional.
If you’re a DES daughter or think you may have been exposed to DES in utero and have experienced harmful side effects, you may also want to consider your legal options. DES victims can fill out a complaint form for a DES attorney to review here.
You’ve Heard of Thalidomide Babies, what about DES Babies?
February 21st, 2012. By AbiK
It’s a startling reality that DES Mothers, DES Daughters and DES Sons live with—that not many people know about or have heard about the pregnancy drug DES—diethylstilbestrol—or the harmful effects its had on both mothers who took the drug, and children who were exposed to the drug in utero.
DES was prescribed during the 1940’s to the 1970’s to expectant mothers who were at risk for miscarriage or pre-term delivery. The drug, however, was found to potentially cause clear cell adenocarcinoma (CCA), a rare form of vaginal and cervical cancer; reproductive tract structural changes such as a T-shaped uterus; complications in pregnancy such as ectopic (tubal) pregnancy and pre-term delivery; and infertility.
DES finally had its FDA approval revoked in 2000 (it had been recommended that doctors not prescibe it in 1971)—but not until potentially millions of mothers and children had been negatively affected by it. And those children are now between forty and seventy years of age or so—some of whom have lived through infertility or miscarriages of their own with out knowing that the reason could be connected to exposure to DES.
And that begs the question: why do so few people know about DES and the damage its caused? DES seems to be in the shadows of thalidomide—so much so that it’s been referred to as the ‘silent thalidomide’ in the press.
There are some possible reasons why, of course.
First, thalidomide was never approved for use in the US. DES was approved which perhaps made it appear less pernicious—after all, ‘my doctor prescribed it’.
Second, thalidomide babies could not help but evoke the collective sympathy and outrage of all who had seen a picture of an innocent newborn with severe birth defects such as missing or shortened arms or other severe disfigurement. There are no alarming pictures of DES victims—only faces; and those faces look just like you or me.
And finally, thalidomide, by its horrific nature, wove its way into popular culture—probably the most familiar evidence of this for many is the Billy Joel lyric from “We Didn’t Start the Fire”.
Diethylstilbestrol (DES), however, has potentially affected many more women in the US—and their children and perhaps their children’s children (aptly called ‘DES Third Generation’)—than thalidomide and is in some ways a much more heinous drug in that DES is the one that slipped through the proverbial cracks—the one that the FDA didn’t put the brakes on—soon enough.
Perhaps you now know about DES. But more importantly, do you know if you were on DES or exposed to it?
Archive by Category
- Accidents (24)
- Airlines (9)
- Asbestos Mesothelioma (262)
- Automotive (25)
- Celebrity (14)
- Class Action (84)
- Complaints/Comments (15)
- Consumer Fraud (84)
- Contest (2)
- Court of Public Opinion (5)
- Crazy Sh*t Lawyers See (61)
- Criminal Law (4)
- Defective Products (111)
- DePuy ASR Hip Recall (2)
- Discrimination (22)
- Drugs/Medical (248)
- Elder Care Abuse (4)
- Emerging Issues (462)
- Employment (54)
- Environment (52)
- Financial (28)
- Food Illness (15)
- Human/Civil Rights (4)
- Insecurities (5)
- Insurance (16)
- Intellectual Property (16)
- Internet/E-commerce (19)
- lawsuits (161)
- Lawyers (20)
- Lawyers Giving Back (43)
- Lex Levity (10)
- Personal Injury (106)
- Pleading Ignorance (53)
- Real Estate (2)
- Recall (6)
- Scam (3)
- Securities (13)
- Settlement (81)
- Tort Reform (2)
- Totally Tortelicious (81)
- Veterans (11)
- Whistleblower (9)